Chemical Forums
General Forums => Comments for Staff and Comments from Staff => Topic started by: curiouscat on April 07, 2016, 10:13:18 AM
-
Am I doing something wrong? The rendering of bicyclo structures seems ugly in SMILES. Any tricks?
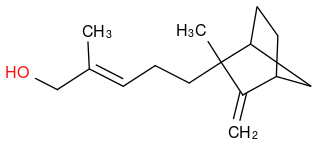
Compare that to the Wikipedia rendering, which seems like the way this thing is most often rendered in books etc. More pleasing to the eye. Unless I'm overlooking a technical difference?
(https://www.chemicalforums.com/proxy.php?request=http%3A%2F%2Fi.imgur.com%2FjHx4sYO.png&hash=8200904663547d68537f1e16af1affabae0560df)
-
SMILES rendering is very random. I seem to recall a recent thread about resonance in phenols in which the phenol group was orientated different ways depending on other substituents ( http://www.chemicalforums.com/index.php?topic=85383.0 ). You may be able to get a better (or nicer) image if you use a different string but no guarantees.
