Post by: bisilara on December 05, 2016, 04:55:23 AM
1) Is Epichlorohydrin carbonate the same as Chloromethyl ethylene carbonate?
(because Chloromethyl ethylene carbonate has a pure sample. If they are the same, then I can use Chloromethyl ethylene carbonate for calibration instead of unavailable Epichlorohydrin carbonate)
2) If they are not the same, can I produce my own Epichlorohydrin carbonate and calibrate it? how can I do this? I am really stuck! any help at all will worth more than a gold
Thank you in advance
Post by: sjb on December 05, 2016, 05:07:26 AM
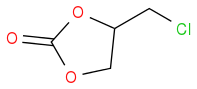 and
and 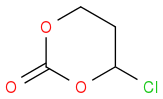
Post by: kriggy on December 05, 2016, 05:24:11 AM
Post by: bisilara on December 05, 2016, 06:07:47 AM
Thank you
Post by: bisilara on December 05, 2016, 06:11:02 AM
I somehow fail to see how the 6 membered compound could be called epichlorhydrine carbonate. Maybe it would be helpful, if OP posted the structure he/she wants to make and then we can talk
I looked at the link but it doesn't give me an English explanation of anything...pls pardon me, I only peak english
Post by: sjb on December 05, 2016, 06:51:30 AM
@ Sjb, thank you, I don't think the 2 structures are the same. so Epichlorohydrin carbonate is definitely not the same as Chloromethyl ethylene carbonate. Ok, how then do I calibrate Epichlorohydrin carbonate to be used as a pretend pure standard for my experiment?
Thank you
Assuming the structures are right, then these are isomers and have the same MW but are likely to have different retention times in HPLC, for instance. How are you planning on showing that you have formed product?
Post by: bisilara on December 05, 2016, 07:33:05 AM
@ Sjb, thank you, I don't think the 2 structures are the same. so Epichlorohydrin carbonate is definitely not the same as Chloromethyl ethylene carbonate. Ok, how then do I calibrate Epichlorohydrin carbonate to be used as a pretend pure standard for my experiment?
Thank you
Assuming the structures are right, then these are isomers and have the same MW but are likely to have different retention times in HPLC, for instance. How are you planning on showing that you have formed product?
This is interesting, like I mentioned, I have only just had one month through my studies, I haven't thought about the performance of individual products in GC analysis. But I think this is the more reason I need to get a pure sample to work with. Another literature I have seen has used both Epichlorohydrin carbonate as a synonym of Chloromethyl ethylene carbonate. Product performance in GC is another phase of my studies I'm planning in the new year
Post by: mjc123 on December 05, 2016, 08:06:05 AM
Post by: sjb on December 05, 2016, 10:45:07 AM
Post by: bisilara on December 05, 2016, 12:14:58 PM
Hmm, further searching around reveals http://www.chemindustry.com/chemicals/02287747.html - maybe the pubmed link is incorrect. Sorry for leading down a blind alley.
That's ok, you're only trying to help. The link below shows that Epichlorohydrin carbonate is used interchangeably Chloromethyl ethylene carbonate both having the same MW. This is really doing my heading
1) http://www.chemindustry.com/chemicals/0427142.html
2) http://www.guidechem.com/reference/dic-59020.html
3) http://www.lookchem.com/Carbonic-acid-cyclic-3-chloropropylene-ester/
3) http://www.lookchem.com/Carbonic-acid-cyclic-3-chloropropylene-ester/
Post by: AWK on December 05, 2016, 07:19:55 PM
Quote
both having the same MWAlmost 200 years organic chemists know isomers.
Post by: bisilara on December 05, 2016, 10:07:57 PM
You use commercial names. They may be ambiguous sometimes.Quoteboth having the same MWAlmost 200 years organic chemists know isomers.
Thank you for your reply, going forward, are these 2 compounds the same or not? Epichlorohydrin carbonate & Chloromethyl ethylene carbonate
Thank you
Post by: Borek on December 06, 2016, 02:46:38 AM
are these 2 compounds the same or not? Epichlorohydrin carbonate & Chloromethyl ethylene carbonate
As AWK told you, commercial names are ambiguous and are not guaranteed to be accurate. It may even happen that different companies use the same name for different compounds. So it is not possible to answer your query, as the only viable answer is: they can be the same, they can be not the same.
The only way to check is to use correct IUPAC names and to contact the company producing samples, asking them for clarification about structure.