Post by: theanonymous on March 25, 2013, 06:10:54 PM
(https://www.chemicalforums.com/proxy.php?request=http%3A%2F%2Fi11.photobucket.com%2Falbums%2Fa179%2Fslourg%2FNUMBER5NEW_zps12b6ad32.png&hash=b5562c7fbf346fbcac31b82bae985f494867b38b)
From my notes in class, this is what I have:
On the first ester equivalent, the alpha proton is removed creating a nucleophilic carbon of the enolate ion. This nucleophilic alpha-carbon attacks the electrophilic carbonyl carbon of the second ester equivalent which throws up the pi bond into a lone pair on the oxide oxygen to form the tetrahedral which intermediate, then comes back down to "spit out" the alkoxide ion which quickly deprotonates the "carbonyl sandwiched" alpha proton. This leaves behind an enolate ion and the alcohol. The acid workup protons the enolate ion at carbon eventually. (May go through tautomerization.)
Species A:
![CC[CH-]C(=O)OCC](https://www.chemicalforums.com/SMILES/2e60c5d160127239e59b.png)
This is wrong :(
I have one more submission left for species A.
Species B:
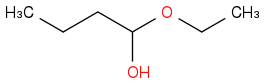
This is wrong too :(
Post by: theanonymous on March 25, 2013, 06:19:19 PM
(https://www.chemicalforums.com/proxy.php?request=http%3A%2F%2Fi11.photobucket.com%2Falbums%2Fa179%2Fslourg%2FHELPPLEASE_zps49ae87b1.png&hash=78d7f89ece95608a7557dff59f715431f3ea516f)
This is a similar problem
Question 9)
(https://www.chemicalforums.com/proxy.php?request=http%3A%2F%2Fi11.photobucket.com%2Falbums%2Fa179%2Fslourg%2FHELPPLEASE2_zps6909cd6e.png&hash=92d59c86f3b0668ce4721f005104dda1994c21da)
I completely messed up on this one
Question 11)
(https://www.chemicalforums.com/proxy.php?request=http%3A%2F%2Fi11.photobucket.com%2Falbums%2Fa179%2Fslourg%2FNUMBER11_zps9ac75c86.png&hash=99ef8d133dc811a8fc127ac742d348bee0e3891f)
This one looked similar to 6 and 5 but I STILL got it wrong :'(
Post by: theanonymous on March 25, 2013, 07:50:30 PM
Post by: discodermolide on March 25, 2013, 08:14:34 PM
question 11 is an aldol condensation.
question 6 we have done already and has nothing to do with the other reactions.
Post by: theanonymous on March 25, 2013, 08:29:13 PM
questions 5, and 9 are Claisen condensations http://en.wikipedia.org/wiki/Claisen_condensation (http://en.wikipedia.org/wiki/Claisen_condensation)
question 11 is an aldol condensation.
question 6 we have done already and has nothing to do with the other reactions.
For 5, is species A this then?
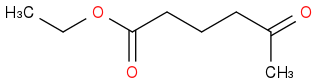
Post by: discodermolide on March 25, 2013, 08:40:07 PM
Post by: theanonymous on March 25, 2013, 09:05:06 PM
No that is not species A. The proton alpha to the carbonyl of one ester attacks the carbonyl of the other ester.
Is the answer
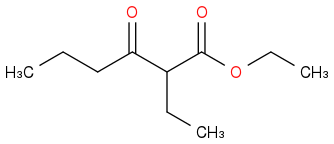 ?
?
Post by: discodermolide on March 25, 2013, 09:12:48 PM
Post by: theanonymous on March 25, 2013, 09:16:36 PM
That is correct.
:D!!!!!!!
Post by: theanonymous on March 25, 2013, 09:17:53 PM
That is correct.
Oh no :(
It says it's wrong...
Post by: discodermolide on March 25, 2013, 09:20:47 PM
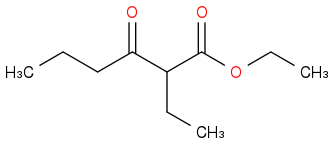
I suppose you could also get this
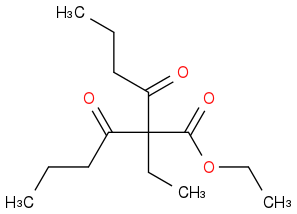
as the middle hydrogen of the first product is then the most acidic one.
Post by: theanonymous on March 25, 2013, 09:23:27 PM
Well the condensation of two equivalents of this ester should give
Idk :(
I've used 3/3 submissions for that one...
So I suppose species B isn't
![CC[O-]](https://www.chemicalforums.com/SMILES/3dc1977fac763bad358b.png) ?
?
Post by: discodermolide on March 25, 2013, 09:26:48 PM
I think B could be ethoxide.
Post by: theanonymous on March 25, 2013, 09:30:25 PM
Sorry I added a bit to my last post.
I think B could be ethoxide.
Species B isn't ethoxide :(
I hate Webassign...
3/3 submissions used for species B...
No hope for the last one.
I have to wait until tomorrow morning at 9:45 AM to see the answers/if your answers were close :(
What about number 6? You said that it's not like the other reactions?
Post by: discodermolide on March 25, 2013, 09:34:49 PM
Let me think about number 6.
Post by: discodermolide on March 25, 2013, 09:40:46 PM
Post by: theanonymous on March 25, 2013, 09:45:13 PM
Sorry I was referring to one of your previous posts. The images are a bit small on my screen and with my eyesight!
Let me think about number 6.
Ok!
Post by: theanonymous on March 25, 2013, 09:47:03 PM
So which ester undergoes a Claisen condensation? And why?
The first one because on the left, there are 2 alpha H protons that can be attacked by a base to make that carbon nucleophilic, which will then be able to attack the second equivalent of the ester.
Post by: discodermolide on March 25, 2013, 09:52:15 PM
Post by: theanonymous on March 25, 2013, 09:55:03 PM
I think it is the first one. The reason the second one doesn't react is probably steric congestion. The anion of the second one is on a tertiary carbon, and to react it must produce a sterically crowded quaternary carbon. This is certainly not likely.
Sooo...for part A, i do the Claisen reaction for the first ester?
Post by: discodermolide on March 25, 2013, 09:56:44 PM
Post by: theanonymous on March 25, 2013, 10:00:47 PM
Yes. :o
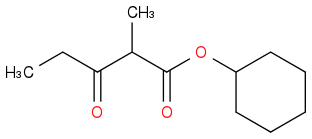
I submitted this... :(:(:( 1/3 submissions...and it's wrong...
aklbfsajksdfkljdfsjkn
I don't even know what to say.
Post by: discodermolide on March 25, 2013, 10:02:21 PM
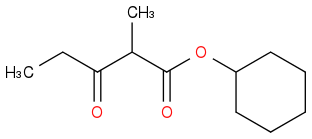
Post by: theanonymous on March 25, 2013, 10:03:23 PM
That's what I would say is correct.
They marked it as wrong though :(
???
Post by: discodermolide on March 25, 2013, 10:09:36 PM
These questions are somewhat misleading as to the reagents used. The acid work up should just protonate everything. I wonder if the acid work up is also hydrolysing the ester in the product to give the acid which then decarboxylates???? Meaning that
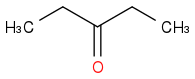
is the product they want?
Post by: theanonymous on March 25, 2013, 10:13:07 PM
Now I am at a loss as to what they want here.
These questions are somewhat misleading as to the reagents used. The acid work up should just protonate everything. I wonder if the acid work up is also hydrolysing the ester in the product to give the acid which then decarboxylates???? Meaning that
is the product they want?
:(:(
I'll try it. But I have no idea what they want either...
I mean if you can't figure it out then my class's average for this HW assignment is gonna be really really low :(
Because I doubt anyone else in my class can figure this out on their own.
Post by: discodermolide on March 25, 2013, 10:17:54 PM
As I wasn't in your class I can't really comment on the material you were given.
I'm sorry I don't seem to be much use tonight :'(
Post by: theanonymous on March 25, 2013, 10:18:16 PM
(https://www.chemicalforums.com/proxy.php?request=http%3A%2F%2Fi11.photobucket.com%2Falbums%2Fa179%2Fslourg%2FANSWER-1_zps89cfbdbe.png&hash=48ea48254e5de42e0fdd9b3e3630ad07e2f62f08)
Is that what we have to do?
Post by: theanonymous on March 25, 2013, 10:20:05 PM
http://answers.yahoo.com/question/index?qid=20090414141221AAsklSv
What does C6H5-O-C(=O)-CH(CH3)-C(=O)C2H5 look like?
Post by: discodermolide on March 25, 2013, 10:22:30 PM
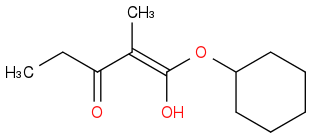
This is also in equilibrium with the keto form and the other enolate.
Post by: theanonymous on March 25, 2013, 10:24:50 PM
You mean they want the enolate as the product?Oh...so are you saying it wouldn't make a difference? :(
This is also in equilibrium with the keto form and the other enolate.
Post by: discodermolide on March 25, 2013, 10:29:47 PM
Post by: theanonymous on March 25, 2013, 10:31:53 PM
Google Keto-enol tautomerisation.Ohh...:(
Post by: theanonymous on March 25, 2013, 10:32:30 PM
Ohh...:(
So they are essentially the same...
Today's been a long day...
these organic chemistry questions are really discouraging.
Ok well...moving on from this question I guess.
Do you know the answer to question 9?
Post by: discodermolide on March 25, 2013, 10:36:14 PM
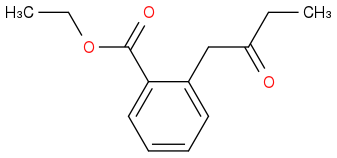
Post by: theanonymous on March 25, 2013, 10:39:12 PM
Yes, I know the answer!
Awesome! :o
That one is correct!
Thanks!!!
What about Number 11...did I get it right this time?
(https://www.chemicalforums.com/proxy.php?request=http%3A%2F%2Fi11.photobucket.com%2Falbums%2Fa179%2Fslourg%2FNUMBER11NEW_zpsd6fbe025.png&hash=24eeb80dd7829e113ad2eabef5a6735efa1c3e1f)
Post by: discodermolide on March 25, 2013, 10:43:19 PM
See if you can work it out otherwise I will draw a scheme.
Post by: theanonymous on March 25, 2013, 10:45:26 PM
No your answer is wrong. This is an aldol condensation.
See if you can work it out otherwise I will draw a scheme.
Oh no :(
I've done it twice..
Post by: theanonymous on March 25, 2013, 10:45:55 PM
No your answer is wrong. This is an aldol condensation.
See if you can work it out otherwise I will draw a scheme.
Oh no :(
I've done it twice..
Can you show me?
Post by: discodermolide on March 25, 2013, 10:48:54 PM
In the meantime I will draw a scheme
Post by: discodermolide on March 25, 2013, 11:03:09 PM
Post by: theanonymous on March 25, 2013, 11:10:14 PM
Here is my scheme:Awesome!!
That structure is correct :D
Thanks!!!
Post by: theanonymous on March 25, 2013, 11:14:02 PM
I have no idea what the answer is but...lemme upload it real quick
Post by: theanonymous on March 25, 2013, 11:16:32 PM
(https://www.chemicalforums.com/proxy.php?request=http%3A%2F%2Fi11.photobucket.com%2Falbums%2Fa179%2Fslourg%2FCRAZYQUESTION2_zps14bb524c.png&hash=2b9c7ff3a1e212017cca564582944de5d3ebe038)
Post by: theanonymous on March 25, 2013, 11:19:07 PM
(https://www.chemicalforums.com/proxy.php?request=http%3A%2F%2Fchemistry.umeche.maine.edu%2FCHY431%2FBasics%2FCyto-Taut.gif&hash=66f20aa04de76b90ee489518044c478c90542e9d)
Post by: theanonymous on March 25, 2013, 11:24:46 PM
Thanks again discodermolide!! For putting up with me and these wretched problems xD
Ill try to figure out #5 and 6 tomorrow morning before 9:45 AM..if not, oh well.
I'm gonna head out..night!
Post by: discodermolide on March 25, 2013, 11:30:38 PM
Here is my answer to the tautomer question.
Post by: theanonymous on March 26, 2013, 01:47:12 PM
The structures were right - but what I forgot to do was deuterate the Hydrogen by the methyl group for both of them...
The structure I got for Species A was actually supposed to be Species C.
And it looks like this:
Question 5)
Species A:
(https://www.chemicalforums.com/proxy.php?request=http%3A%2F%2Fi11.photobucket.com%2Falbums%2Fa179%2Fslourg%2FSPECIES_A_zps1d573396.png&hash=d8b6af7a822f7fdb855558ff91439b106e1f4b1a)
Species B:
(https://www.chemicalforums.com/proxy.php?request=http%3A%2F%2Fi11.photobucket.com%2Falbums%2Fa179%2Fslourg%2FSPECIES_B_zps6a1e3998.png&hash=80affc5ebbf3dac40511514a3d39979ff4647764)
Species C:
(https://www.chemicalforums.com/proxy.php?request=http%3A%2F%2Fi11.photobucket.com%2Falbums%2Fa179%2Fslourg%2FSPECIES_C_zps4d600e6e.png&hash=8d05e911c329ba20fe0cf99dce744eaaabb4e83e)
Question 6)
(https://www.chemicalforums.com/proxy.php?request=http%3A%2F%2Fi11.photobucket.com%2Falbums%2Fa179%2Fslourg%2FNUMBER_6ANSWER_zps5850464a.png&hash=d229d01564446f98de0a4d5a9bee10db02e89a49)
:D!
I feel like I'm slowly getting better at Organic Chem :P
Post by: discodermolide on March 26, 2013, 02:01:36 PM