It’s been a long time since I have done any organic chemistry, so I apologise in advance if I have said anything crazy…
I just need to know whether there is a simple way to conjugate one of the pyridine groups from the TPEN molecule (a metal chelator; see below), to a terminal carboxyl group of another compound, either directly or through a linker molecule. I also need the TPEN to remain functional as a weak Ca
2+ or Mg
2+ chelator once conjugated.
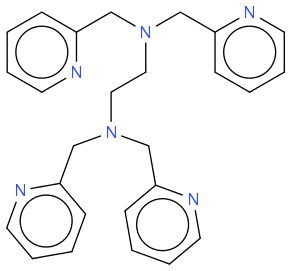
TPEN
I do quite a bit of copper catalysed “click chemistry” between azide/alkyne terminated compounds. So it would be awesome if it were possible to add either of those groups to the pyridine/carboxyl groups. My only worry with this would be if the TPEN permanently chelated the copper catalyst and was no longer sensitive to Ca
2+/Mg
2+….
In short I'm trying to achieve something like this:

+
![[*:0]C(=O)O](https://www.chemicalforums.com/SMILES/218a8882aeceff56000e.png)
=
![c1ccc(nc1)CN(CCN(Cc1ccccn1)Cc1ccccn1)Cc1cc([*:0])ccn1](https://www.chemicalforums.com/SMILES/9ae3896d85894db4cd8e.png)
TPEN + "R" linked carboxyl = Linked TPEN-"R" composite
It doesn't matter where or how the secondary molecule "R" binds as long as TPEN is still functional.
I would be very grateful if anyone could help me out with this, I’ve been stuck for a while.
All suggestions welcome!
Thanks in advance.
