Post by: curiouscat on June 21, 2013, 03:20:40 AM
Do I get free chlorine like so?
H2O2 (aq.) + HCl (aq. :rarrow: H2O + HOCl (aq.)
HOCl (aq.) + HCl (aq.) ::equil:: Cl2 :spinup: + H2O
We use a H2O2 / HCl mix for making a chlorohydrin and I'm confused as to what species actually takes part in the reaction.
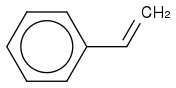 :rarrow:
:rarrow: ![c1ccccc1C(O)C[Cl]](https://www.chemicalforums.com/SMILES/91ff4f777d9bf995732e.png)
Is it the hypochlorite ion that reacts?
HClO (aq.) ::equil:: OCl− + H+
Or nascent Chlorine? Or something else?
If it matters, HCl and H2O2 are both ~30% and temp. is between 60 to 90 C.
If someone has a guess about the mechanism, I'd love to hear it! I've a guess that the Chloronium Ion is involved somewhere but not sure.
Post by: opsomath on June 21, 2013, 10:12:50 AM
Here is an interesting discussion of the reaction, which apparently leads mostly to HCl-catalysed decomposition to O2 at low concentrations, but yields substantial chlorine concentrations in the interim.
https://sites.google.com/site/unusualchemistry/halogen-hydrogen-peroxide-interactions
Post by: curiouscat on June 22, 2013, 03:14:41 AM
It's interesting to see a reaction as simple as H2O2 + HCl seems to still have quite a bit of confusion about it! :)
Post by: Borek on June 22, 2013, 04:21:36 AM
Post by: curiouscat on June 22, 2013, 04:44:08 AM
Usually it means reaction is far from simple - many possible pathways, which one dominates depends on the details, so there is no single answer.
Right. "seemingly simple" is what I meant.
What confuses me even more is I'm not even sure what species I really need for the reaction!
Post by: curiouscat on June 22, 2013, 09:05:20 AM
H2O2 + 2 H+ + 2 Cl- :rarrow: Cl2 + 2 H2O
H2O2 + Cl2 :rarrow: O2 + 2 H+ + 2 Cl-
If this were dominant in my reaction I should see a disparity in the liquid mass balance right? Even if all the nascent Cl reacted and none was lost as Cl2 gas, the evolved O2 is almost the entire mass fraction of the peroxide.
But I don't see this mass deficit.
What gives?
Post by: camptzak on June 22, 2013, 08:43:39 PM
how else could it go other than protonation of a peroxide oxygen, and chlorine substitution releasing water?
Post by: opsomath on June 24, 2013, 10:43:39 AM
I do think that this:
Quote
Even if all the nascent Cl reacted and none was lost as Cl2 gas, the evolved O2 is almost the entire mass fraction of the peroxide.
is incorrect. Given the equations from the link, for every mole of chlorine that is produced, one mole of peroxide goes directly to form water. Since the O2 producing reaction consumes a mole of chlorine, the very most you could lose to oxygen is half the peroxide.
Since we get "moderate steady bubbling that last for several hours" I'm assuming that the second reaction is relatively slow (reasonable, since it's the oxidation of something that's already pretty highly oxidized) and that the solution contains a high concentration of latent and dissolved chlorine as Cl2, HOCl, and more complex chlorine oxide species. This is a pretty reasonable conclusion since, after all, this reaction mixture is effective at your halohydrin reaction!
Do you have access to a UV-Vis spectrometer? You could place some of the reaction mixture in a cuvette and monitor the absorption over time. I think molecular Cl2 absorbs at 325-350 nm. You could even get some kinetics for the reaction this way.
I assume the styrene reaction is biphasic. That complicates things considerably, because it is essentially an extraction. Cl2 is extremely polarizable and nonpolar, so it would get schlepped into the styrenic phase (making it unavailable to decompose peroxide further) and virtually ensuring it would react with a molecule of styrene. Then (here's the clever bit) the chloronium ion which formed would be polar enough to re-partition back into the aqueous peroxide phase, where it would most likely react with a water molecule. (Reaction with a peroxide molecule would give you some side products including alpha-chloroacetophenone).
tl;dr Whoever designed your reaction is a smart cookie.
Post by: curiouscat on June 26, 2013, 02:52:02 AM
It is an interesting scenario, isn't it?
Indeed! :)
Quote
is incorrect. Given the equations from the link, for every mole of chlorine that is produced, one mole of peroxide goes directly to form water. Since the O2 producing reaction consumes a mole of chlorine, the very most you could lose to oxygen is half the peroxide.
Right you are. I'll check my material balance more carefully. Let's see if I can observe this. Though there's a bit of evaporation too that makes it hard to tease out.
Quote
Do you have access to a UV-Vis spectrometer? You could place some of the reaction mixture in a cuvette and monitor the absorption over time. I think molecular Cl2 absorbs at 325-350 nm. You could even get some kinetics for the reaction this way.
I'll try! Good ideal. So far quantification was on a GC.
Quote
I assume the styrene reaction is biphasic.
Yes. That's where our rate determining step lies I think.
Quote
Whoever designed your reaction is a smart cookie.
Is that a bug or a feature though? :) The bi-phasic part, I mean.
One idea I've been toying with is to get the darn phases to emulsify somehow to see if that will enhance rates. Good idea or bad, you think?
Post by: Archer on June 26, 2013, 04:03:16 AM
This fits with the formation of hypochlorous acid (HOCl) in the reaction between water and chlorine. The hypochlorous acid is what reacts with your alkene.
Post by: opsomath on June 26, 2013, 09:48:16 AM
Quote
Is that a bug or a feature though? The bi-phasic part, I mean.
One idea I've been toying with is to get the darn phases to emulsify somehow to see if that will enhance rates. Good idea or bad, you think?
I think it's a feature. Essentially your oxidation reaction (Cl- + H2O2) takes place between extremely polar things, but produces an extremely nonpolar byproduct, Cl2. This stuff naturally partitions into your styrene phase and reacts there, producing a polar intermediate which naturally partitions back into the other phase.
Emulsifying might increase the boundary surface area allowing the Cl2 to diffuse into the other phase better. But if it's already mixed well, I wouldn't expect it would matter much.
Post by: magician4 on June 26, 2013, 07:40:10 PM
hence, i would consider standard epoxidation followed by acid-catalysed ringopening with subsequent chloride-addition as another plausible pathway...
... just like we know it from the standardsystem formic acid / H2O2 (with alpha-hydroxy formic esters resulting)
regards
Ingo
Post by: curiouscat on June 27, 2013, 01:08:40 AM
hence, i would consider standard epoxidation followed by acid-catalysed ringopening with subsequent chloride-addition as another plausible pathway...
Interesting. Can you elaborate what you mean by "standard epoxidation"? Do you have a particular route in mind? Since industrially epoxides are very often made via halohydrins I find your counter-pathway intriguing.
Post by: opsomath on June 27, 2013, 10:26:08 AM
Post by: magician4 on June 27, 2013, 10:28:32 AM
(https://www.chemicalforums.com/proxy.php?request=http%3A%2F%2Fupload.wikimedia.org%2Fwikipedia%2Fcommons%2Fthumb%2F5%2F5a%2FEpoixd_Butterfly.png%2F300px-Epoixd_Butterfly.png&hash=f9620584d3d2829682b2b16177c970b5ec53727d)
(from: http://commons.wikimedia.org/wiki/File:Epoixd_Butterfly.png )
depending on the general reaction conditions those epoxides can either be isolated, or will undergo acid-catalyzed ringopening, resulting in alpha-hydroxy-esters as next product (which upon further treatment in aq. systems will undergo hydrolysis, resulting in trans-1,2 dioles)
(https://www.chemicalforums.com/proxy.php?request=http%3A%2F%2Fupload.wikimedia.org%2Fwikipedia%2Fcommons%2Fthumb%2F9%2F97%2FCyclohexanediolSynthesis.svg%2F400px-CyclohexanediolSynthesis.svg.png&hash=8d9a9f88477a7790089eb05bb5a14114bc6b5a77)
(from: link (http://upload.wikimedia.org/wikipedia/commons/thumb/9/97/CyclohexanediolSynthesis.svg/400px-CyclohexanediolSynthesis.svg.png))
(though shown as base-catalyzed ester cleavage here: this might well happen with acid catalysts, too )
very reactive olefines, like for example styrene systems, don't require the organic acid as a by-catalyst for epoxidation
(https://www.chemicalforums.com/proxy.php?request=http%3A%2F%2Fupload.wikimedia.org%2Fwikipedia%2Fcommons%2Fthumb%2Fc%2Fc8%2FAlgar-Flynn-Oyamada-Reaktion-Neu.svg%2F420px-Algar-Flynn-Oyamada-Reaktion-Neu.svg.png&hash=8ee37b8a107032191b448babad9f663272e51f6c)
(from: link (http://upload.wikimedia.org/wikipedia/commons/thumb/c/c8/Algar-Flynn-Oyamada-Reaktion-Neu.svg/420px-Algar-Flynn-Oyamada-Reaktion-Neu.svg.png) )
(note that the initial epoxide-formation is left out both in the upper as as in the lower row, though it clearly must take place)
regards
Ingo
Post by: curiouscat on June 27, 2013, 10:30:41 AM
What are the main byproducts of the reaction? That should shed some light on the mechanism.
Benzaldehyde and
![c1ccccc1C([Cl])C[Cl]](https://www.chemicalforums.com/SMILES/a133c8518700a0947296.png) seem the major by-products that we see. Some very little
seem the major by-products that we see. Some very little 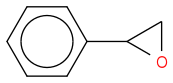 too.
too. There's some stuff that we haven't identified yet.
Post by: magician4 on June 27, 2013, 11:02:44 AM
beats me how that might be formed from styrene, esp. as I would expect it to become further oxidized towards benzoic acid under those conditions... **)
regards
Ingo
*)
from the redox-reaction H2O2, HCl --> H2O , Cl2 discussed previously
**)
it would require ozone (or something that behaves like it) to result this product, and I don't see how we should get this form the setup
Post by: curiouscat on June 27, 2013, 11:21:54 AM
beats me how that might be formed from styrene, esp. as I would expect it to become further oxidized towards benzoic acid under those conditions... **)
**)
it would require ozone (or something that behaves like it) to result this product, and I don't see how we should get this form the setup
I'm not sure myself! Good point.
I remember @opsomath mentioned Benzoic Acid in a related post. May be he has an idea how.
Would Benzoic Acid partition out into the aqueous phase? I have only noticed benzaldehyde as a by-product.
Post by: magician4 on June 27, 2013, 11:31:51 AM
Quote
Would Benzoic Acid partition out into the aqueous phase?benzoic acid is very poorly soluble in neutral / acid waterbased systems (2.9 g/L , 25°C) , though its solubility is acceptable under basic conditions (as the corresponding anion, that is)
hence, depending on your very concentrations, it MIGHT partition out more or less completely, yes
regards
Ingo
Post by: Archer on June 27, 2013, 11:44:55 AM
Hypochlorous acid would not be ionised at low pH and is formed in equlibrium with Cl2 when water and chlorine are mixed.
The same reaction happens if you mix chlorine with an alcohol but, obviously, you make the ether insted of the hydroxyl group.
Post by: opsomath on June 27, 2013, 12:00:11 PM
Post by: Archer on June 27, 2013, 12:11:57 PM
Post by: Archer on June 27, 2013, 12:17:37 PM
"The mechanism of HOX addition is electrophillic, with initial attack by the positive halogen end of the HOX dipole. Following Markovnikov's rule, the positive halogen goes to the side of the double bond that has more hydrogens. The resulting carbocation reacts with the OH- or H2O to give the product."
Post by: magician4 on June 27, 2013, 12:26:03 PM
Quote
Hypochlorous acid would not be ionised at low pH and is formed in equlibrium with Cl2 when water and chlorine are mixed.agreed, but...
under acid conditions named equilibrium will be shifted far far towards Cl2/H2O
additionally , how the relatively weak and at the same time low-concentrated HOCl should win the race vs. Cl2 with respect to the attack of the doublebond I don't see
@ opsomath
Quote
I thought the mechanism for the chlorine/alcohol addition was solvolysis of the chloronium ion, not formation of alkyl hypochlorite....witch opens another pathway to the known product
regards
Ingo
Post by: curiouscat on June 27, 2013, 12:40:02 PM
under acid conditions named equilibrium will be shifted far far towards Cl2/H2O
additionally , how the relatively weak and at the same time low-concentrated HOCl should win the race vs. Cl2 with respect to the attack of the doublebond I don't see
Exactly what I wondered when I first saw this proposal! But I assure you we get fairly decent selectivity to the halohydrin.
Could it be that the Cl2 does win, the chlorinated product forms, and it subsequently hydrolysed in situ to the halohydrin by the HCl present?
PS. Thanks all of you for the very interesting discussion BTW. Appreciate your insights! Being an Engineer, the minutiae of mechanisms isn't my forte. :)
Post by: Archer on June 27, 2013, 12:47:13 PM
under acid conditions named equilibrium will be shifted far far towards Cl2/H2O
additionally , how the relatively weak and at the same time low-concentrated HOCl should win the race vs. Cl2 with respect to the attack of the doublebond I don't see
To quote March again (same page) "HOCl is often generated in situ by the reaction between water and Cl2"
Don't forget that Cl2 has a much weaker dipole than HOCl so in this reaction there would be far less "Cl+" with Cl2 than with HOCl.
I am basing my statements on data I have collected and books that I have read and can reference.
If you wish to poropose an alternative mechanism which is in contrast to experimental data and published theories then I can't really win a debate so can't offer much more to this discussion.
Post by: Archer on June 27, 2013, 12:50:18 PM
Could it be that the Cl2 does win, the chlorinated product forms, and it subsequently hydrolysed in situ to the halohydrin by the HCl present?
This is unlikely as it would be attacking a very hindered carbon to give your product. The primary alcohol would be favoured if this were the case.
Post by: magician4 on June 27, 2013, 01:10:39 PM
Quote
Could it be that the Cl2 does win, the chlorinated product forms, and it subsequently hydrolysed in situ to the halohydrin by the HCl present?I take it that we should allow for several parallel mechanisms to occur: both the chlorine-pathway (by 1,2 dichlorphenylethane) and the epoxide-pathway (by the very epoxide being observed) are supported by your findings of by-products, and the addition of HOCl of course is an option, too
question is: which of the possible pathways is the most relevant ?
as part of the chlorine-additon pathway:
for the "Cl2 wins" - scenario both your addition / hydrolysis mechanism as as (in my opinion more plausible) the direct reaction of the initial chloronium-ion*) with water will lead to your product
so many ways lead to Rome....
regards
Ingo
*)
chlorination under these conditions is an electrophilic addition of Clδ+-Clδ-, and will take place as a 2-step reaction, where in the second step the benzylic cation is attacked by whatever nucleophile wins the race: water or chloride in this case are likely candidates
however, benzylhalogenides (i.e. the chloride-anion is the winner) are known to be moisture-sensitive with respect to an X- /OH- replacement, so the hydrolysis-pathway also is plausible
Post by: magician4 on June 27, 2013, 01:36:15 PM
Quote
To quote March again (same page) "HOCl is often generated in situ by the reaction between water and Cl2"I take it that you will agree, that the degree of HOCl formation is highly different if I compare the reaction of chlorine with water (March) to the reaction of chlorine with aqueous hydrochloric acid (our setup)
Quote
Don't forget that Cl2 has a much weaker dipole than HOCl so in this reaction there would be far less "Cl+" with Cl2 than with HOCl.I wouldn't want to derive relative reaction velocities from this single aspect: there's so much more to consider, let alone "concentration"
Quote
I am basing my statements on data I have collected and books that I have read and can reference.so do all of us, i take it, to the best of their knowledge
Quote
If you wish to propose an alternative mechanism which is in contrast to experimental data (...)the experimental data reported sofar by curiouscat explicitly describes the epoxide and the dichloride as known by-products (which your favoured pathway can't explain at all)
if anything, these products support the alternative mechanisms you so fiercely denote as "in contrast to known data"
again: March is talking about water/chlorine, not HCl/chlorine as matrix!
this IS a big difference!
Quote
This is unlikely as it would be attacking a very hindered carbon to give your product. The primary alcohol would be favoured if this were the case.don't forget that we're talking styrene here: in this system the benzylic position is the more stable to harbour the cation
--> I don't agree with your analysis at this point
regards
Ingo
Post by: Archer on June 27, 2013, 01:55:11 PM
I would love the oportunity to test these theories experimentally. I think we all agree that this warrants further investigation.
I love this forum, it's good to be challenged on mechanistic chemistry. I would not get this anywhere else. No one where I work tends to discuss such things and my friends generally switch off as soon as I start talking chemistry :)
Post by: curiouscat on June 27, 2013, 02:19:35 PM
I would love the oportunity to test these theories experimentally. I think we all agree that this warrants further investigation.
I do have a experimental rig going on. So will be glad to try anything that comes up.
If there are any ideas for an experiment, do post and I'll try to run them. So far we've only been trying to optimize our product distribution in a pretty brute-force approach.
Post by: Archer on June 27, 2013, 03:10:30 PM
I would chlorinate styrene n DCM with Cl2 and try to form your product in conc. HCl
Third I would heat your product in conc HCl to see if the epoxide forms.
The only thing I hate more than being wrong is being wrong twice! I do a lot of this chemistry and all I care about is the product. How I got there is academic so It's nice have a different theory to consider, thanks guys!
My interest for my research in this would be the β methyl styrene and the effect on relative configuration. Using your method (i.e cis vs trans).
Post by: curiouscat on June 30, 2013, 12:09:43 AM
I would look at careful acidification of hypochlorite solution, and visa verse, see how that works out.
What did you mean by this one? I dose acid / H2O2 and the substrate together; do you mean changing the ratios? The hypochlorite solution gets generated in situ, if at all.
Post by: Archer on June 30, 2013, 06:30:54 AM
Expt 1 I would add the substrate to sodium hypochlorite solution and slowly add HCl (no H2O[2.) This would make hypochlorous acid and maybe your product.
Expt 2 add substrate to HCl and add sodium hypochlorite slowly, the equilibrium should favour Cl2 see what the outcome of this reaction is.
In addition to the other recommendations I made previously (treating phenyl-1,2-dichloroethane and your product with HCl) you could also see what happens when you treat styrene oxide with HCl and/or with HCl & H2O2
Others may consider these a complete waste of time, this is just what I would (rightly or wrongly) try to develop a better understanding the potential processes which are going on. These can all be done on a relatively small scale but really you have to question whether all this is an effective use of your time.
Post by: magician4 on June 30, 2013, 12:44:49 PM
having done some mechanistic studies in the past whilst doing my PhD, I strongly would recommend to define the very scientific question you are about to research before going into experimental details
we see from the literature and the side products that at least 3 pathways might occur which are worthy of further investigation
furthermore , we know form the byproduct "benzaldehyde" that at least one complete set of interactions between them educts / intermediates is beyond even our professional imagination for the time being
:rarrow: I would be highly astonished to learn that from a handfull of well chosen experiments the whole situation comes apart in a way, that will reveal the truth of the matter like the sun rising in the morning
there's more to it , much more, my guts tell me...
so, what is the goal of the endeavour?
for-real improving of a technical process?
... and if not, i.e. if we're talking a scientific analysis of the different pathways etc., this will require a lot of time and money to shine a substantial light on the fines of the action
remember: even those seemingly simple systems like "oxygene / hydrogene - reaction leading to water" know dozends of pathways and species, and to the best of my knowledge still is not fully completed in mechanistic studies yet
regards
Ingo
Post by: Archer on June 30, 2013, 05:53:03 PM
Unfortunately I can't describe how I know this in too much detail as this would violate the forum rules but put simply the bonding electrons between the benzylic carbon and the next carbon in the chain are transferred to the oxidant.
Post by: magician4 on June 30, 2013, 06:36:46 PM
Quote
Benzaldehyde is a common side reaction (sometimes the main product) of oxidation of certain benzyl alcohols with hypochlorous acid and could also happen with Cl2, although I have never tried.to the best of my knowledge, you would need a Ar-CH(OH)-R structure to achieve this, with R being either H or -COOH
any pathway including an explanation to the benzaldehyde findings hence would have to explain the intermediate formation of mandelic acid first (or any alternative path to get rid of the terminal =CH2 fragment, exchanging it for hydrogens)
regards
Ingo
Post by: Archer on July 01, 2013, 03:49:28 AM
to the best of my knowledge, you would need a Ar-CH(OH)-R structure to achieve this, with R being either H or -COOH
any pathway including an explanation to the benzaldehyde findings hence would have to explain the intermediate formation of mandelic acid first (or any alternative path to get rid of the terminal =CH2 fragment, exchanging it for hydrogens)
Indeed Ar-CH(OH)-R is what you need but this could be the result of hydrolysis of any epoxide formed or even just the product of the reaction.
As you will no doubt be aware the general method for the oxidation of the vicinal diol to benzladehyde is to use periodate, I have never tried to use hypochlorite but this may work to a lesser extent yielding benzaldehyde.
There is an R group of Ar-CH(OH)-R which is known to oxidise to benzaldehyde with hypochlorite, unfortunately I cannot describe this owing to forum rules. I have contacted a moderator to validate this but it is documented in the literature.
As you have already stated the benzylic cation is very stable so benzylic alcohols will lose electrons to an oxidising agent (such as hypohalite / hypohalous acid) leaving benzaldehyde as the product to a greater or lesser extent.
Post by: curiouscat on July 01, 2013, 07:38:07 AM
There is an R group of Ar-CH(OH)-R which is known to oxidise to benzaldehyde with hypochlorite, unfortunately I cannot describe this owing to forum rules. I have contacted a moderator to validate this but it is documented in the literature.
Thanks for all your help @Archer! I wouldn't want to tax you to write anything more down but just in case, I doubt it would violate any Forum Rules if you did.
IMHO, those caveats are for people seeking easy homework solutions whereas here you and @magician seem to have a very interesting discussion going on (and at least I'm finding it very educational) and I doubt the Mods would mind your elaborating any more.
Just my 2 cents. Of course, the Mods can correct me if I am wrong.
Post by: magician4 on July 01, 2013, 11:00:36 AM
besides: I would be eager to learn which groups other than -H , -COOH are sensible to HOCl in the way we're discussing here, coming from styrene in a plausible pathway, as the literature I did look up regarding this didn't mention it.
but then again: who knows all the literature? so pls. be so kind
regarding the discussion in a broader way: periodate will split vic. diols, yes
on the other hand: IMHO no chlorine-based species will do so (else this much cheaper substance would have been used for this purpose in the SOP's of the old days)
... and even if we had traces of iodine in the HCl , i would believe in the formation of periodate even less than i believe in the formation of high concentrations of HOCl under the conditions described.
epoxides are ok with me, but then we would need findings of the diol, the mandelic aldehyde and the mandelic acid in the trace section , too , aside from just benzaldehyde (which, I would expect, is even more sensible to oxidation than mandelic aldehyde)
as I said: looking into this system seriously, I would expect a whole bunch of interesting things to happen and a lot of work to be done
and by the end of the day, that's all I wanted to mention: this question is none answered in a New York minute
regards
Ingo
Post by: curiouscat on July 02, 2013, 12:02:40 AM
so, what is the goal of the endeavour?
for-real improving of a technical process?
Goal: To make as much halohydrin as I can from given quantity of styrene (as fast as I can).
This boils down to (IMHO):
1. Minimising pathways leading to (a) benzaldehyde (b) dichloride (c) styrene polymerization
2. Increasing rate constants for primary reaction by optimising temperature / finding a catalyst
3. Finding conditions that promote hydrolysis of the dichloride to the halohydrin.
Post by: discodermolide on July 02, 2013, 12:08:43 AM
Post by: curiouscat on July 02, 2013, 12:30:03 AM
What is the purity of your styrene?
Thanks @disco.
It's commodity Styrene Monomer, not extra pure or anything. (Lyyondell Basel I think.)
I believe 96%+ is the spec. but I will double check. It comes with inhibitor added at ppm levels.
Can I ask, if you had a particular problem in mind? If I guess, thinking that the Benzaldehyde is an oxidised impurity from the RM?
Post by: discodermolide on July 02, 2013, 01:28:17 AM
Post by: curiouscat on July 02, 2013, 01:36:16 AM
I just wondered if benzyl alcohol was an impurity. How much benzaldehyde do you see?
~5%. In some rare cases a little more.
Post by: discodermolide on July 02, 2013, 01:52:56 AM
I wonder if you are getting some C=C cleavage, don't ask me how, but with the oxidant you are using who knows?
Post by: curiouscat on July 02, 2013, 01:58:13 AM
That's quite a lot of benzaldehyde.
I wonder if you are getting some C=C cleavage, don't ask me how, but with the oxidant you are using who knows?
Agreed! :)
Luckily, or unluckily, I care less about the actual pathway but if you have any tips ( catalyst, conditions, excess reagents, addition order etc.) that'd have a shot at maximizing my selectivity towards the halohydrin do let me know!
Post by: discodermolide on July 02, 2013, 02:44:08 AM
Post by: curiouscat on July 02, 2013, 03:16:20 AM
What's your order of addition?
All three at once in almost exact stoichiometric ratios. Starting fluid in reactor is an excess of weak spent acid.
Post by: discodermolide on July 02, 2013, 03:28:16 AM
Post by: curiouscat on July 02, 2013, 03:31:52 AM
Why not add the oxidant to the styrene?
I'll try. Reaction temp. being 60°C plus I was afraid high, hot styrene conc. were invitations for runaway or polymerization byproducts. That was the reason I was diluting down the reaction mix. But only speculating.
Quote
Or better add styrene to the oxidant, then you may have more control?
That's worth a shot too.
Post by: discodermolide on July 02, 2013, 03:41:12 AM
Post by: curiouscat on July 02, 2013, 04:06:51 AM
So add the styrene to the oxidant. This is a two phase reaction mixture? If so then stirring is an important factor.
It is indeed.
Can you think of any co-solvents etc. that might ease the two-phase mass transfer issues? A phase transfer catalyst?
Post by: discodermolide on July 02, 2013, 04:09:57 AM
Or *Ignore me, I am impatient* up the temperature, and make sure the process is addition controlled, i.e. never an excess of styrene, almost complete conversion by the time the addition is complete. You've got great heat capacity in the water, so you should be OK thermally.
Post by: discodermolide on July 02, 2013, 04:24:48 AM
Post by: curiouscat on July 02, 2013, 05:52:19 AM
So add the styrene to the oxidant.
Will try. Thanks.
PS. A mix of H2O2 + HCl (both ~30% w/w) at ~70°C is not crazy is it? Just checking. Earlier this went in together as it reacted but now I'll need a lot of it. (if I'm adding Styrene in slowly to oxidant)
Any obvious incompatibility? Outside of the general nastiness of peroxides and acids, of course. That's for granted.
Post by: discodermolide on July 02, 2013, 06:54:14 AM
Post by: curiouscat on July 02, 2013, 07:03:24 AM
Test it out in the lab first, you will soon see. Or have you access to a DSC?
Oh yes, of course. Wasn't meaning I'd try this on pilot / production scale direct.
DSC would have been great, but no, don't have one.
Post by: discodermolide on July 02, 2013, 09:28:07 AM
Post by: Archer on July 02, 2013, 10:26:26 AM
I reduced some phenacyl bromide (as I had some in stock) to the alcohol very crudely to see if I could then oxidise it to benzaldehyde with NaOCl in acetic acid. I couldn't!, in fact the major product looks like ring chlorination!)
The reduction was quite effortless though (no benzaldehyde), the only side reaction was some epoxide which may be because I didn't neutralise during the work up I just removed the methanol with a stream of nitrogen as it was only very small scale (10 mg in 1 ml).
Post by: magician4 on July 02, 2013, 11:23:24 AM
I think that toluene is a by-product in styrene manufacture. This may be the source of the benzaldehyde, if you have any toluene in your styrene?Styrene industrial grade typically is like 99,5% +
mostly it is made from ethylbenzene via cat. dehydration, and the ethylbenzene is made from benzene and ethylene
:rarrow: no major source for typically up to 5% benzaldehyde (from toluene) here
a typical analysis of industrial grade styrene will show like 0.02% benzaldehyde
http://www.lookchem.com/Product_1967917/CasNo_100-42-5/Styrene.html#.UdLpSdhGQgk
:rarrow: there should be a real pathway from styrene to benzaldehyde: this is not coming from impurities
@ curiouscat :
70°C with 30% H2O2 looks pretty harsh to me on the H2O2
maybe this is enough to provoke (minor) thermal decomposition already: H2O2 :rarrow: H2O + O.
with molecular oxygen around, this might rearrange to form ozone, which then will split the styrene
:rarrow: to test for this theory, you might wish to either lower temperature or work with protective gas, and see if reduction of temperature (less oxygen-radicals) or reduction of elementary oxygen will reduce the typical amount of benzaldehyde thereby
regards
Ingo
Post by: curiouscat on July 02, 2013, 01:03:26 PM
@ curiouscat :
70°C with 30% H2O2 looks pretty harsh to me on the H2O2
maybe this is enough to provoke (minor) thermal decomposition already: H2O2 :rarrow: H2O + O.
Probably. I see some bubbling in the H2O2 as it is in the dosing funnel and especially where it meets the reaction mix.
Quote
with molecular oxygen around, this might rearrange to form ozone, which then will split the styrene
:rarrow: to test for this theory, you might wish to either lower temperature or work with protective gas, and see if reduction of temperature (less oxygen-radicals) or reduction of elementary oxygen will reduce the typical amount of benzaldehyde thereby
Great idea. I will try that.
Post by: curiouscat on July 04, 2013, 12:45:01 AM
Could you make your product by chemical reduction (NaBH4) of phenacyl chlroide?
Probably could if it were a lab synthesis.
Commercially it won't make sense (I think). NaBH4 and phenacyl chloride are both more expensive I suspect than HCl + H2O2. Even Styrene is a bulk commodity chemical. Even with perfect yields versus the imperfections of my status quo, I doubt it's gonna work.
I doubt the economics would work out. But I could be wrong.
Post by: curiouscat on August 21, 2013, 09:07:29 AM
@ curiouscat :
70°C with 30% H2O2 looks pretty harsh to me on the H2O2
maybe this is enough to provoke (minor) thermal decomposition already: H2O2 :rarrow: H2O + O.
with molecular oxygen around, this might rearrange to form ozone, which then will split the styrene
:rarrow: to test for this theory, you might wish to either lower temperature or work with protective gas, and see if reduction of temperature (less oxygen-radicals) or reduction of elementary oxygen will reduce the typical amount of benzaldehyde thereby
Just thought I'd post an update regarding my follow up on one of @magician's ideas:
Tried reducing the reaction temperature. This screws up the selectivity. Apparently as T reduces I make more and more of the dichloride. (As an aside, is this something people would expect, even in hindsight, from the underlying chemistry?)
So, one productive result was that I'm now working at slightly higher T and this gives a better halohydrin : dichloride (i.e. product:byproduct) ratio.
Thanks @magician! :)