Post by: zsinger on July 19, 2014, 09:47:16 AM
I was helping an undergraduate student, and I ran into a synthesis which I just could not figure out for some reason. Maybe I am having a brain fart, but see what y'all think. The ONLY idea I had was starting with trans-2-phenyl-1-cyclohexanol, modifying it perhaps, and doing some sort of EAS. Im sorry for the lack of a pic, so I will attempt to name it and describe it best I can, and maybe one of the nice mods will draw it for me. I read the formatting and have no idea how to draw this one still:
I believe the name would be 1-Phenyl-Cyclohexene (phenyl ring attached to a cyclohexene -->The Phenyl ring is attached directly to one of the carbons participating in the double bond)
Any help appreciated.
-Zack
Post by: zsinger on July 19, 2014, 09:52:59 AM
Zack
Post by: Dan on July 19, 2014, 10:09:08 AM
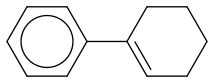
Any restrictions as to starting materials?
zsinger - benzene is a very poor Friedel-Crafts nucleophile. You could do something like that with anisole etc. But benzene will not work well. Then you have the classic overalkylation issue as well. The product would need further manipulation to install the alkene.
The target screams "cross coupling" to me!
Post by: zsinger on July 19, 2014, 10:47:54 AM
-Zack
Post by: zsinger on July 19, 2014, 10:57:09 AM
-Zack
Post by: discodermolide on July 19, 2014, 12:43:07 PM
Post by: zsinger on July 20, 2014, 02:26:25 PM
Post by: orgopete on July 21, 2014, 07:09:16 AM
Post by: zsinger on July 21, 2014, 11:38:30 AM
-Zack
Post by: orgopete on July 21, 2014, 11:54:25 AM
Post by: zsinger on July 22, 2014, 12:56:54 PM
-Zack
Post by: orgopete on July 22, 2014, 03:03:14 PM
So make the alcohol, then dehydrate as per usual E1 with H2SO4 and Δ?
-Zack
That is what I would think. That route seems simple and very much like the chemistry of an intro ochem course. Obviously if the problem restricts you to five carbons or less, it may be different. If it is a problem in a chapter of Grignards, then definitely.
Post by: zsinger on July 23, 2014, 06:04:36 PM