Post by: Sir_Goat on March 30, 2015, 08:25:53 AM
I've used methanol as solvent, added my base, potassium tert-butoxide, and allowed it to equilibriate for 10-15 minutes. Then I added the 2,4-pentanedione
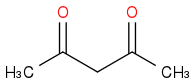 which was left again for 5 minutes followed by the addition of 4-nitrobenzyl bromide
which was left again for 5 minutes followed by the addition of 4-nitrobenzyl bromide=O)C=C1](https://www.chemicalforums.com/SMILES/e88eade90607e89d2292.png)
The mixture was then left stirring overnight and the solvent then removed via rotary evaporation. The product was analysed with NMR in CDCl3 and it seems to have not attached in the centre but at the end like so
=O)C=C1)=O)=O](https://www.chemicalforums.com/SMILES/0a727624c899eb7c55cd.png)
Why has this happened? I added a 1:1 base mixture and surely the hydrogens on the 3- postion should be more acidic?
To make matters worse I'm struggling to recreate my results (basically no reaction occurs on some attempts).
Any suggestions as to why it reacts like it does or why I'm struggling to recreate my results would be much appreciated. :)
Post by: TheUnassuming on March 30, 2015, 09:34:48 AM
Post by: Sir_Goat on March 30, 2015, 11:46:12 AM
However, I have just realized that most of them used non-protic solvents as oppose to protic ones like methanol. Maybe this is what is causing me the problem?
Post by: TheUnassuming on March 30, 2015, 12:41:33 PM
That said the general reaction scheme you've shown is done in a number of different solvents with many different bases. So if the method you posted first doesn't work or is finicky, try some other common systems like NaH/THF or carbonate/acetone.
Post by: clarkstill on March 30, 2015, 01:20:16 PM
Post by: orgopete on April 02, 2015, 07:33:41 PM