Post by: njoyeux on November 07, 2022, 02:54:38 PM
I have a question regarding the resonance of pyracyclene.
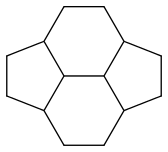
If I’m correct, pyracyclene has four resonance forms:
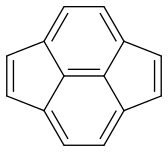
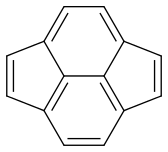
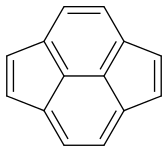
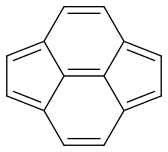
Now my problem is estimating the dominance of each form. Are they equal or is there one which is more dominant?
This is what I’ve got so far:
Rules for estimating stability of resonance structures
- The greater the number of covalent bonds, the greater the stability since more atoms will have complete octets.
- The structure with the least number of formal charges is more stable.
- The structure with the least separation of formal charge is more stable.
- A structure with a negative charge on the more electronegative atom will be more stable.
- Positive charges on the least electronegative atom (most electropositive) is more stable.
- Resonance forms that are equivalent have no difference in stability and contribute equally (eg. Benzene).
Pyracyclene has the same carbon atoms, the same number of covalent bonds and a formal charge of zero. I don’t see any difference in stability either. So it seems that all forms are equivalent.
Is this correct?
Post by: Enthalpy on November 07, 2022, 07:18:23 PM
Post by: njoyeux on November 08, 2022, 08:19:43 AM
The pyracyclene molecule has two symmetries: up/down and left/right. You can see that all symmetries for all forms are present.
Post by: Orcio_87 on November 09, 2022, 04:03:39 PM
Post by: Enthalpy on November 10, 2022, 06:05:26 AM
The symmetry of the first form is the first form itself.My bad.
Post by: Enthalpy on November 10, 2022, 06:09:41 AM
It looks like it fullfils the Huckel's rule, so all four structures should be equal.I don't grab this logic. Does Hückel's rule tell anything more than "aromatic"? In 1,2-dichlorobenzene, I don't expect both forms to be equally probable.
Post by: njoyeux on November 11, 2022, 07:59:29 AM
It looks like it fullfils the Huckel's rule, so all four structures should be equal.I don't grab this logic. Does Hückel's rule tell anything more than "aromatic"? In 1,2-dichlorobenzene, I don't expect both forms to be equally probable.
I don't grab the logic either.
Plus, on the following research paper pyracylene is described as a bordeline case of aromaticity.
https://pubs.acs.org/doi/pdf/10.1021/ja012541%2B (https://pubs.acs.org/doi/pdf/10.1021/ja012541%2B)
What does that tell us?