Post by: Bertrand on January 04, 2020, 04:26:46 AM
I have some problems with making a connection in order to reach the final product of my synthesis.
I want to connect and amide an aldehyde thought a nucleophilic addition in order to get an N, O-alcohol product (Hemiaminal).
could you please give some advice? I have been trying to make that connection for a month using several papers but none of them lead me to the product.
Thank you very much in advance for your propositions
Post by: rolnor on January 04, 2020, 01:14:32 PM
Post by: Bertrand on January 06, 2020, 04:13:46 PM
Yes, the target compound is pretty unstable. You will find attached the reaction scheme.
OC(C)(C)C)C(=O)Nc1ccccc1C(N)=O](https://www.chemicalforums.com/SMILES/00a60e23231edbcb24ea.png) +
+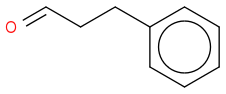 going to
going toOC(C)(C)C)C(=O)Nc1ccccc1C(=O)NC(O)CCc2ccccc2](https://www.chemicalforums.com/SMILES/2068ad7b2c1188c7458d.png)
(mod edit convert structures to SMILES for inline representation)
Post by: kriggy on January 07, 2020, 07:01:52 AM
https://pubs.acs.org/doi/10.1021/ol4031155
TBH, I dont think you can isolate your compound. There are cases of stable hemiaminals with free OH groups but thats AFAIK only in ring systems.
Post by: Bertrand on January 07, 2020, 10:38:49 AM
I tried the synthetic mentioned in the paper but the synthesis did not work.
Post by: rolnor on January 07, 2020, 02:51:05 PM
Post by: Bertrand on January 07, 2020, 03:29:20 PM
Post by: rolnor on January 07, 2020, 06:30:16 PM
Post by: rolnor on January 08, 2020, 03:57:17 PM
Post by: Bertrand on January 08, 2020, 05:55:43 PM
The compound has not been synthesized before. It has been isolated from a plant
Post by: Bertrand on January 08, 2020, 06:02:51 PM
Here is a paper in which the N, O-alcohol bond is made.
Post by: hypervalent_iodine on January 08, 2020, 07:14:43 PM
Hello!
The compound has not been synthesized before. It has been isolated from a plant
This seems...questionable. The compound you've shown looks to be quite unstable. If the isolation has been published, I would be very curious to look at the paper. I have seen examples of terpene derived hemiaminals, but those were able to be synthesised and shown to be stabilised through intramolecular H bonding (https://pubs.acs.org/doi/abs/10.1021/ol025558l). I'm not sure your target would be able to benefit from the same level of interactions.
Post by: rolnor on January 08, 2020, 11:34:25 PM
Post by: kriggy on January 09, 2020, 01:15:13 AM
Based on all those publications shown in the thread I think it could be made because the aminals they prepare also dont have much hydrogen bonding to stabilize the aminal. They mostly do alcoholysis afterwards so maybe protecting the OH could work to make it more stable. There is a important note in one of the papers:
"The crude hemiaminals are unstable towards prolonged contact with water; therefore, a rapid workup is recommended!" however, they do aq. extractions...
OP: are you sure you have dry glassware and solvents? Maybe thats the problem.¨Also, could be nice to see what conditions did you try
Post by: Bertrand on January 09, 2020, 02:09:41 AM
I tried the conditions mentioned in this paper https://doi.org/10.1002/ejoc.201500655.
I even tried the synthetic route mentioned in this one (https://pubs.acs.org/doi/10.1021/ol4031155) basing on the synthesis of compound 7 which is quite similar to my target compound in terms of steric hindrance.
Post by: Bertrand on January 09, 2020, 02:48:17 AM
Please, I need your help to find out a good synthetic route of compound 4 of this paper https://doi.org/10.1016/j.bbrc.2016.09.041
Thanks in advance
Post by: rolnor on January 09, 2020, 02:41:53 PM
Post by: Bertrand on January 09, 2020, 03:04:15 PM
I am still searching for how to make it. Please, all your suggestions will, of course, be welcome.
Post by: rolnor on January 09, 2020, 05:20:54 PM
I have the same thoughts. The major issue will probably be making the hemiaminal and attach the thiol group (starting from carbon 7 to the thiol group)
I am still searching for how to make it. Please, all your suggestions will, of course, be welcome.
I think a good start would be to use 2-azidobenzamide and try to add it to the thiolaldehyde. The azido group can be reduced with PPh3 to amine after this. I dont think you can have a Boc-protected amino acid, the deprotection will cleave the hemiaminal. Maybe a Fmoc is better, the hemiaminal should be stable to base like piperidine wich is used for Fmoc-deprotection. The first step with the azidobenzamide reacting with thiolaldehyde would be great to study directly with NMR, the aldehyde should dissapear and you get something close ta a acetal-peak. If you run TLC then use a system with base like triethylamine or use aluminunoxide instead if silica wich is acidic to avoid hydrolysis of the hemiaminal. One bad thing is the thiol wich smells veeeery bad i guess.
Post by: Bertrand on January 10, 2020, 02:13:05 AM
I think instead of using the smelling thiol group, maybe I should protect it with trityl to also avoid any reactivity.
I am going to try your suggestion
Post by: rolnor on January 10, 2020, 02:02:38 PM
Post by: Bertrand on January 10, 2020, 04:25:28 PM
you are right... to avoid any acid SEM-group is better than trityl