Post by: egilbreth on March 30, 2020, 04:04:47 PM
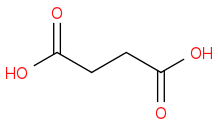 :rarrow:(Br2,hv)
:rarrow:(Br2,hv)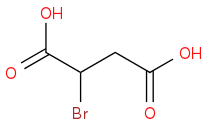 :rarrow:(NaOH, Δ)
:rarrow:(NaOH, Δ) 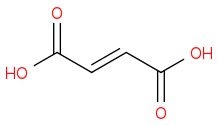 :rarrow:(H2SO4,H2O)
:rarrow:(H2SO4,H2O)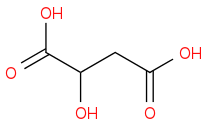 :rarrow:(CrO3, H2SO4)
:rarrow:(CrO3, H2SO4) 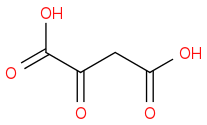 :rarrow:(Δ)
:rarrow:(Δ)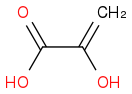 +
+ 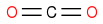
_______________________________________________________________________________________________
 :rarrow: (H2,Ni)
:rarrow: (H2,Ni) 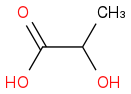 :rarrow:(H2SO4, Δ)
:rarrow:(H2SO4, Δ) 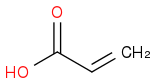 :rarrow: (H2,Ni)
:rarrow: (H2,Ni) 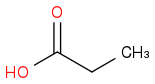
I think it's possible but I was unsure if there would be problems when trying to brominate the original acid and if adding a ketone to split it like that would even be possible. Any feedback is much appreciated
Post by: rolnor on March 30, 2020, 04:26:53 PM
https://en.wikipedia.org/wiki/Pyruvic_acid
One thing that could be very difficult is the work-up, you have very polar compounds and H2SO4 and cromium-salts, have you thought about that? Also I see risc of decarboxylation in some steps.
Post by: egilbreth on March 30, 2020, 04:43:13 PM
Also would PCC work better than the H2SO4 and cromium-salt or does that run into the same issue because of the polar compound and acid
Post by: Babcock_Hall on March 30, 2020, 05:26:53 PM
Post by: wildfyr on March 30, 2020, 09:05:48 PM