Post by: theanonymous on April 03, 2013, 07:27:44 PM
Also, what does the structure for the following look like?
A) octane-3,5-diamine
B) N-butyl-N-methylcyclohexylamine
C) Cation of N-methylpentylammonium iodide
Post by: theanonymous on April 04, 2013, 12:05:56 AM
Post by: discodermolide on April 04, 2013, 12:25:12 AM
Surely you can have a go at drawing the structures from the others, it's all in a name.
Give it a go and post your answers.
Post by: theanonymous on April 04, 2013, 12:57:43 AM
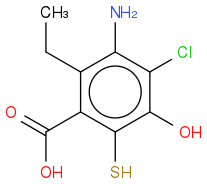
The name of this compound is 3-amino-4-chloro-2-ethyl-5,6-dihydroxybenzoic acid, thanks to ChemDraw.
Surely you can have a go at drawing the structures from the others, it's all in a name.
Give it a go and post your answers.
Ok!
Post by: theanonymous on April 04, 2013, 01:02:09 AM
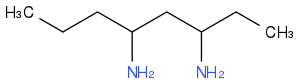
Post by: theanonymous on April 04, 2013, 01:05:22 AM
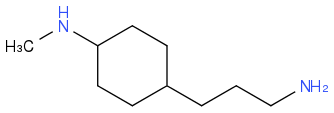
??
Post by: theanonymous on April 04, 2013, 01:07:31 AM
Post by: discodermolide on April 04, 2013, 01:20:01 AM
For the second compound it says N-Butyl N-Methyl………
So your N-Methyl is correct, what about the N-Butyl?
Note the chain in your structure only has 3 carbon atoms not 4 as butyl has!!!
Post by: theanonymous on April 04, 2013, 07:58:49 AM
OK with the octane,3,5 diamine.
For the second compound it says N-Butyl N-Methyl………
So your N-Methyl is correct, what about the N-Butyl?
Note the chain in your structure only has 3 carbon atoms not 4 as butyl has!!!
I just submitted this structure...it says that it's wrong :(
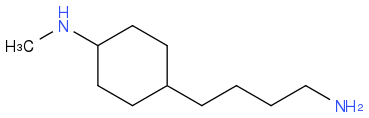
Post by: sjb on April 04, 2013, 08:01:01 AM
OK with the octane,3,5 diamine.
For the second compound it says N-Butyl N-Methyl………
So your N-Methyl is correct, what about the N-Butyl?
Note the chain in your structure only has 3 carbon atoms not 4 as butyl has!!!
I just submitted this structure...it says that it's wrong :(
How many amines are there in your name, and in your structure as drawn?
Post by: sjb on April 04, 2013, 08:02:11 AM
The name of this compound is 3-amino-4-chloro-2-ethyl-5,6-dihydroxybenzoic acid, thanks to ChemDraw.
Surely you can have a go at drawing the structures from the others, it's all in a name.
Give it a go and post your answers.
There's only one phenol...
Post by: theanonymous on April 04, 2013, 08:07:58 AM
The name of this compound is 3-amino-4-chloro-2-ethyl-5,6-dihydroxybenzoic acid, thanks to ChemDraw.
Surely you can have a go at drawing the structures from the others, it's all in a name.
Give it a go and post your answers.
There's only one phenol...
Yeah I was gonna say... I submitted '3-amino-4-chloro-2-ethyl-5,6-dihydroxybenzoic acid' but got it wrong.
Should it be 3-amino-4-chloro-2-ethyl-5,6-hydroxybenzoic acid?
Post by: discodermolide on April 04, 2013, 08:08:52 AM
it is,
3-amino-4-chloro-2-ethyl-5-hydroxy-6-mercaptobenzoic acid
Post by: theanonymous on April 04, 2013, 08:09:00 AM
OK with the octane,3,5 diamine.
For the second compound it says N-Butyl N-Methyl………
So your N-Methyl is correct, what about the N-Butyl?
Note the chain in your structure only has 3 carbon atoms not 4 as butyl has!!!
I just submitted this structure...it says that it's wrong :(
How many amines are there in your name, and in your structure as drawn?
Oh...there's supposed to be three, no? because of the 'amine' at the end.
But where would that go?
Post by: discodermolide on April 04, 2013, 08:12:42 AM
Post by: theanonymous on April 04, 2013, 08:18:28 AM
The clue is in the N-Methyl, N-Butyl.Ohhh
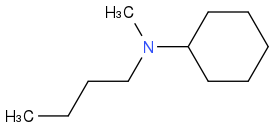
Post by: discodermolide on April 04, 2013, 08:19:51 AM
Post by: theanonymous on April 04, 2013, 08:20:49 AM
One carbon too many in the chain, you have 5 it should be 4.

Post by: discodermolide on April 04, 2013, 08:22:00 AM
Post by: theanonymous on April 04, 2013, 08:24:50 AM
Now what about the last compound?
C) Cation of N-methylpentylammonium iodide
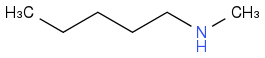
But they want the cation of that?
Post by: discodermolide on April 04, 2013, 08:26:55 AM
----ammonium iodide.
Post by: theanonymous on April 04, 2013, 08:30:06 AM
Yes what is a cation and what an anion in this case?
----ammonium iodide.
Oops i meant...
Would it just be ammonium?
Post by: discodermolide on April 04, 2013, 08:42:23 AM
![CCCCC[NH2+]C.[I-]](https://www.chemicalforums.com/SMILES/2c31193274b7013d7fb6.png)
so the cation is the molecule without the I-
![CCCCC[NH2+]C](https://www.chemicalforums.com/SMILES/3922bb7d06562dffeafa.png)
Post by: theanonymous on April 04, 2013, 08:43:34 AM
This you mean?
Oh is that the answer?
Post by: discodermolide on April 04, 2013, 08:44:12 AM
Post by: theanonymous on April 04, 2013, 08:51:21 AM
See above
Would it be [NH3+]I?
I don't get it...
Post by: discodermolide on April 04, 2013, 08:54:04 AM
![CCCCC[NH2+]C](https://www.chemicalforums.com/SMILES/3922bb7d06562dffeafa.png)
Note the NH+2
Post by: theanonymous on April 04, 2013, 08:55:18 AM
The cation is the positively charged part.
Note the NH+2
![C[NH3+]](https://www.chemicalforums.com/SMILES/dca1f15c319134dcfa9f.png) ?
?
Post by: discodermolide on April 04, 2013, 08:59:05 AM
The cation is the positively charged part.
Note the NH+2?
No, the answer I posted, it's the entire molecule positively charged but without the iodide
Post by: theanonymous on April 04, 2013, 09:01:29 AM
Ohh!The cation is the positively charged part.
Note the NH+2?
No, the answer I posted, it's the entire molecule positively charged but without the iodide
Thanks! :D