Post by: curiouscat on May 27, 2013, 08:22:24 AM
 with H2O2 + HCl (with heating) one makes
with H2O2 + HCl (with heating) one makes ![c1ccccc1C(O)C[Cl]](https://www.chemicalforums.com/SMILES/91ff4f777d9bf995732e.png) .
. In terms of byproducts which may I expect? My expectations (partially based on literature searches ) are
![c1ccccc1C([Cl])C[Cl]](https://www.chemicalforums.com/SMILES/a133c8518700a0947296.png) and
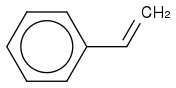
and ![c1c([Cl])cccc1C=C](https://www.chemicalforums.com/SMILES/15ecb31b9b4934f0c857.png) and
and ![c1([Cl])ccccc1C=C](https://www.chemicalforums.com/SMILES/6511ae67a307cae1a5ca.png) .
.But is there anything else that might be major competition? Am I missing any?
Any strategies to minimize these undesired by products.
Post by: orgopete on May 28, 2013, 12:50:47 PM
Post by: opsomath on May 28, 2013, 02:22:00 PM
Not surprised about the aromatic substitution, I think I've seen chlorides + peroxide as a green-chemical chlorinating reagent before. I think with a copper catalyst? Maybe could reduce this if you made sure everything's clean of transition metals.
One other thing; peroxides make radicals, radicals polymerize styrene, or at least cause anti-Markovnikov addition.
Post by: curiouscat on May 28, 2013, 02:32:40 PM
I'm with orgopete, I'd say that over-oxidation is a likely culprit. Benzoic acid?
On further investigation, it seems benzaldehyde was one significant byproduct especially when something goes wrong.
Quote
Not surprised about the aromatic substitution, I think I've seen chlorides + peroxide as a green-chemical chlorinating reagent before. I think with a copper catalyst? Maybe could reduce this if you made sure everything's clean of transition metals.
Thanks. I'll see what I can do. Our plan was test batches in a Glass Lined Reactor.
Quote
One other thing; peroxides make radicals, radicals polymerize styrene, or at least cause anti-Markovnikov addition.
Yes, especially at 100 C preventing Styrene from polymerizing is going to be challenging.
Post by: curiouscat on May 29, 2013, 02:35:29 AM
Or would that be likely to inhibit my desired reaction too? Is the inhibitor action a free radical scavenger? Or....?
Post by: opsomath on May 29, 2013, 08:42:16 AM
Post by: curiouscat on May 29, 2013, 09:58:17 AM
I think that BHT or whatever would probably react with your peroxide directly. I definitely remember running an oxidation of hydroquinone in peroxide at like 70°C...but that was with iodine catalyst. So I guess my answer is, yes, it makes sense to try, but it may not stand up to the peroxide.
Thanks. It's a tricky problem.
Normally styrene is stored below 30 C to avoid polymerization. Especially above 65 C the risk of highly exothermic runaway polymerization is high. And my desired halohydrin formation (based on most patents) is carried from 80 - 100 C.
So essentially I'm operating much above the self-polymerization kickoff temperature.