Post by: Studentofstuff123 on May 31, 2013, 02:59:04 AM
Thank you
(https://www.chemicalforums.com/proxy.php?request=http%3A%2F%2Fs24.postimg.org%2Fi5iz20lp1%2Fs2344353d.png&hash=fea596dbb7851a0797d79202dbf494cbc3edaa7a)
Post by: Babcock_Hall on May 31, 2013, 09:15:38 AM
Post by: Studentofstuff123 on May 31, 2013, 11:35:26 AM
I can't find it in my biochem book though.
Post by: Babcock_Hall on May 31, 2013, 12:16:02 PM
Post by: Studentofstuff123 on June 01, 2013, 03:09:19 AM
I think that what makes it difficult for me is that I see the overall functional group as the combination two common functional groups. By itself, the side chain of an asparagine residue is an amide. If you combine an amine with a hemiacetal derived from a carbohydrate, what do you obtain?
I am not sure. I'm kind of lost on this one. What would be your best guess?
Post by: curiouscat on June 01, 2013, 05:14:18 AM
Post by: billnotgatez on June 01, 2013, 06:08:58 AM
I was wondering if you read forum rules
Post by: Arkcon on June 01, 2013, 10:09:42 AM
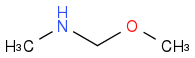
Is that sort of close to it -- ignoring that the rest of the sugar ring has to be called a CH3 (I'm not very good at SMILES yet)? Like everyone else, I see a couple of functional groups, but I don't know a name for this one by itself. Unless the letters for the atoms spell your school's initials, or something. Could that be it?
Post by: DrCMS on June 01, 2013, 10:52:33 AM
Post by: Babcock_Hall on June 01, 2013, 12:06:23 PM
Post by: Kikko on June 02, 2013, 06:10:53 AM
@OP, You must make an attempt; that is a forum rule. However, I will give you a hint. The functional group that connects a base (purine or pyrimidine) to a ribose or deoxyribose in RNA or DNA respectively, is similar.Is it a tertiary amine ?
(but on the first/original question in would be a secondary amine?)
Post by: Dan on June 02, 2013, 09:42:51 AM
Is it a tertiary amine ?
No, look it up: example (http://csbg.cnb.csic.es/mbrole/files/func_groups_recognized_checkmol.pdf) of a list I found on google.
In your original post you highlighted this fragment:
![[*:1]OC([*:2])N[*:3]](https://www.chemicalforums.com/SMILES/cb7a5a48e98acdcf4096.png)
Post by: Kikko on June 02, 2013, 10:12:11 AM
Is it a tertiary amine ?
No, look it up: example (http://csbg.cnb.csic.es/mbrole/files/func_groups_recognized_checkmol.pdf) of a list I found on google.
In your original post you highlighted this fragment:
I dit not ask it :p
But was curious.
Ah yes, I forgot the O.
I guess its a hemiaminal than?
Post by: Studentofstuff123 on June 03, 2013, 03:58:06 AM
Is it a tertiary amine ?
No, look it up: example (http://csbg.cnb.csic.es/mbrole/files/func_groups_recognized_checkmol.pdf) of a list I found on google.
In your original post you highlighted this fragment:
I dit not ask it :p
But was curious.
Ah yes, I forgot the O.
I guess its a hemiaminal than?
After reading the list provided, I'm thinking it could be hemiaminal, but that has 5 R groups. Is it a hemiaminal?
Post by: Dan on June 03, 2013, 04:40:22 AM
After reading the list provided, I'm thinking it could be hemiaminal, but that has 5 R groups. Is it a hemiaminal?
The specific linkage in the red box is a hemiaminal - not sure where you are getting 5 R groups from:
You have in this case:
![[*:1]OC([*:2])N[*:3]](https://www.chemicalforums.com/SMILES/cb7a5a48e98acdcf4096.png)
R1 = sugar ring; R2 = sugar ring; R3 = Asn residue of peptide
More specifically (also considering what is outside the red box) you could call this structure an N-acyl hemiaminal (because R3 is an acyl group).
Based on the core amide structure:
![[*:1]NC(=O)[*:2]](https://www.chemicalforums.com/SMILES/5259a304de973b00d644.png)
and given that R1 = glycosyl, you could call it an N-glycosyl amide. There are a lot of functional groups present, so there is more than one logical way of describing what is present.
Post by: Kikko on June 03, 2013, 05:08:32 AM
After reading the list provided, I'm thinking it could be hemiaminal, but that has 5 R groups. Is it a hemiaminal?
The specific linkage in the red box is a hemiaminal - not sure where you are getting 5 R groups from:
You have in this case:
R1 = sugar ring; R2 = sugar ring; R3 = Asn residue of peptide
More specifically (also considering what is outside the red box) you could call this structure an N-acyl hemiaminal (because R3 is an acyl group).
Based on the core amide structure:
and given that R1 = glycosyl, you could call it an N-glycosyl amide. There are a lot of functional groups present, so there is more than one logical way of describing what is present.
In the PDF file you posted, with the names, there are 5 R groups, I guess thats why he is confused that its an hemiaminal.
On wiki its showing an "easier" structure:
http://en.wikipedia.org/wiki/Hemiaminal
(https://www.chemicalforums.com/proxy.php?request=http%3A%2F%2Fupload.wikimedia.org%2Fwikipedia%2Fcommons%2F8%2F8c%2FHemiaminal-2D-skeletal.png&hash=a9b959a5d34afa1be1a002fd80bc224fdd8c3561)
Mod edit: Image resized. Dan
Post by: Studentofstuff123 on June 03, 2013, 05:25:07 AM
After reading the list provided, I'm thinking it could be hemiaminal, but that has 5 R groups. Is it a hemiaminal?
The specific linkage in the red box is a hemiaminal - not sure where you are getting 5 R groups from:
You have in this case:
R1 = sugar ring; R2 = sugar ring; R3 = Asn residue of peptide
More specifically (also considering what is outside the red box) you could call this structure an N-acyl hemiaminal (because R3 is an acyl group).
Based on the core amide structure:
and given that R1 = glycosyl, you could call it an N-glycosyl amide. There are a lot of functional groups present, so there is more than one logical way of describing what is present.
Hmmm...thank you Dan. I will tell my professor on Monday. I am sure he will ask me how I figured it and will definitley tell him from here because he is way too smart to think I figured this by myself.
He basically challenged everyone in the class to find this and he said NO one has EVER figured it out in 10+ years. I just wanted to see why that was.
I will keep you all posted, thank you.
Post by: Kikko on June 03, 2013, 05:26:01 AM
Is it a tertiary amine ?
No, look it up: example (http://csbg.cnb.csic.es/mbrole/files/func_groups_recognized_checkmol.pdf) of a list I found on google.
In your original post you highlighted this fragment:
Looking back at it, I am not sure where you get your "fragment".
I can understand the R3 group.
But what is R1 and R2 ? One of them has to be the sugar group where N is linked to (and contains the "red O", but the other R?
Isnt Arkcon his figure better?
=>
OK, so ... removing all the rest of the graphic, and drawing it out, (and converting to SMILES so it displays here,)
Is that sort of close to it -- ignoring that the rest of the sugar ring has to be called a CH3 (I'm not very good at SMILES yet)? Like everyone else, I see a couple of functional groups, but I don't know a name for this one by itself. Unless the letters for the atoms spell your school's initials, or something. Could that be it?
Also
@OP, You must make an attempt; that is a forum rule. However, I will give you a hint. The functional group that connects a base (purine or pyrimidine) to a ribose or deoxyribose in RNA or DNA respectively, is similar.And I dont think the functional group that connects a base to a ribose is an hemiaminal
Post by: Dan on June 03, 2013, 06:33:19 AM
Looking back at it, I am not sure where you get your "fragment".
I can understand the R3 group.
But what is R1 and R2 ? One of them has to be the sugar group where N is linked to (and contains the "red O", but the other R?
Isnt Arkcon his figure better?
This is what I did:
Post by: Kikko on June 03, 2013, 07:18:02 AM
Looking back at it, I am not sure where you get your "fragment".
I can understand the R3 group.
But what is R1 and R2 ? One of them has to be the sugar group where N is linked to (and contains the "red O", but the other R?
Isnt Arkcon his figure better?
This is what I did:
Ok, I see what you did, but I am still confused about what R1 and R2 is.
Does R2 refer to the "second" sugar ? (the one attached to the "first" sugar that contains the O that is found in the red square?)
Post by: Arkcon on June 03, 2013, 07:43:54 AM
Post by: Kikko on June 03, 2013, 07:48:31 AM
Well, R1 and R2 are by definition anything, since the O.P. drew a red box to exclude much of the ring. Its a bit confusing, since in the original molecule, what ends up as R1 and R2 are parts of the same ring, but the question isn't about that.
Ok, but if I look at your drawing compared to the one Dan does.. to me those are different molecules...
Thus changing the name of the functional group?
Or?
Post by: Arkcon on June 03, 2013, 07:59:26 AM
Post by: AWK on June 03, 2013, 08:19:20 AM
Post by: Babcock_Hall on June 03, 2013, 09:17:39 AM
Post by: Kikko on June 03, 2013, 10:32:53 AM
It is common to refer to the linkage between a base and the (deoxy)ribose in RNA or DNA as an N-glycoside. However, this is frowned upon in some circles. "...however the use of 'N-glycoside', although widespread in biochemical literature, is improper and not recommended here ('glycosylamine' is a perfectly acceptable term)." One wonders whether the present group might be called a glycosylamide. http://www.chem.qmul.ac.uk/iupac/2carb/33.html
Its getting a bit more complicated..
Now how do you call the structure in DNA that you described: @OP, You must make an attempt; that is a forum rule. However, I will give you a hint. The functional group that connects a base (purine or pyrimidine) to a ribose or deoxyribose in RNA or DNA respectively, is similar.
You would say its a hemiaminal or glycosylamide ?
Because I justed asked around and they told me that what you describe is also called a tertiary amine...
So I am confused!
ALso: if I look up the definition of Glycosidic bond, I see it refers to having a hemiacetal group and this is not the case in DNA so I wonder where they came up with this glycosidic bond.
Post by: Dan on June 03, 2013, 11:47:48 AM
They differ in their level of generality. N-Glycosylamides are a type of N-acetylhemiaminal.
Post by: Babcock_Hall on June 03, 2013, 02:08:31 PM
ALso: if I look up the definition of Glycosidic bond, I see it refers to having a hemiacetal group and this is not the case in DNA so I wonder where they came up with this glycosidic bond.Glycosides are a form of acetal, not hemiacetal.
Post by: Studentofstuff123 on June 05, 2013, 07:11:46 AM
Post by: Kikko on June 06, 2013, 05:26:17 AM
He confirmed that it was indeed correct, hemiaminal ether more specifically
Pretty weird that the biochemistry majors dont know this.
Altough, I guess its more a thing for (organic) chemistry majors.