Post by: limonade on August 12, 2013, 10:47:18 PM
I have drawn out the mechanisms on scratch paper. It makes sense to me. I gave the dehydration products as shown in the picture and it counted me as wrong. I have no idea if it wants the neutral aldo product before it undergoes dehydration. It won't tell me what I did wrong, chemically speaking not program wise, and I would like to know otherwise I just might make the same mistake again, if I have made any. Any help greatly appreciated. Thanks
Post by: discodermolide on August 13, 2013, 02:11:19 AM
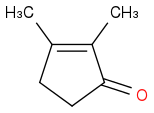
unless it is asking for the product before loss of water. But the aldol condensation is the synthesis of conjugated carbonyl compounds.
Post by: Dan on August 13, 2013, 03:00:02 AM
Post by: limonade on August 13, 2013, 09:22:08 AM
Thank you