Post by: Rutherford on February 16, 2014, 08:06:32 AM
Post by: discodermolide on February 16, 2014, 08:36:05 AM
Both OH and NH are tosylated.
Post by: discodermolide on February 16, 2014, 08:40:59 AM
http://en.wikipedia.org/wiki/4-Dimethylaminopyridine (http://en.wikipedia.org/wiki/4-Dimethylaminopyridine)
Post by: Rutherford on February 16, 2014, 08:52:24 AM
Post by: Rutherford on February 16, 2014, 09:00:52 AM
Post by: discodermolide on February 16, 2014, 09:18:19 AM
Post by: Rutherford on February 16, 2014, 09:46:09 AM
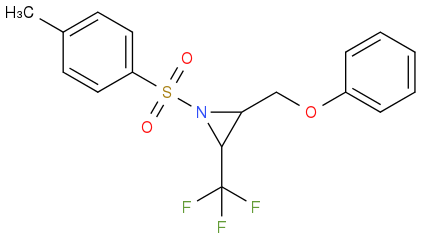
Post by: discodermolide on February 16, 2014, 09:53:13 AM
Please draw the product you think it is.
Post by: discodermolide on February 16, 2014, 09:58:08 AM
Post by: Rutherford on February 16, 2014, 09:58:35 AM
Post by: discodermolide on February 16, 2014, 10:12:22 AM
Note that very occasionally when I made a tosylate and tried to displace it with a nucleophile the reaction didn't work. What happened was that the chloride from triethylamine hydrochloride had already done the job. This happened only with certain compounds, but you should be aware that this side reaction can take place. I avoided this side reaction by using a different base.