Post by: stephanopolous on March 31, 2014, 07:00:04 PM
Here is the problem:
C6H5COH + CH3CN------>EtO-, EtOH-----> (C9H7N)---->H3O+/heat---->
I believe the C9H7N is telling you that's what you have after the first steps, not telling you to add that.
I have the answer (guessed it correctly) but I want to understand why it's that.
Any help would be greatly appreciated. So far all I did was a nucleophilic attack on the aldehyde by the nitrile (electrons on the N) to form an intermediate, but I'm not sure where to go from there.
I looked through all of our power point slides, all of the book chapters we covered, and googled 17,000 different ways that it may come up - nothing.
Post by: discodermolide on March 31, 2014, 07:56:20 PM
I don't think that C9H7N is correct.
Post by: stephanopolous on March 31, 2014, 08:09:41 PM
Post by: discodermolide on March 31, 2014, 08:30:18 PM
The acetonitrile is deprotonated by the base to give a resonance stabilised anion. This attacks the aldehyde to give the product I drew.
You take it from there.
Post by: stephanopolous on March 31, 2014, 08:42:25 PM
Thanks again for drawing that out, that was really nice of you.
Post by: discodermolide on March 31, 2014, 08:43:53 PM
What happens to nitriles when you treat them with acid?
Post by: stephanopolous on March 31, 2014, 08:45:12 PM
Post by: stephanopolous on March 31, 2014, 08:47:23 PM
Post by: stephanopolous on March 31, 2014, 08:48:19 PM
Post by: stephanopolous on March 31, 2014, 08:49:56 PM
Post by: stephanopolous on March 31, 2014, 08:55:22 PM
Post by: discodermolide on March 31, 2014, 09:00:37 PM
The first product is still a nitrile.
The acid hydrolyses it to the carboxylic acid and causes the elimination of water to give the product you drew.
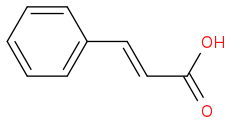
Post by: critzz on March 31, 2014, 09:00:58 PM
In the attachment I drew the full mechanism of the first addition + elemination.
I didn't draw the hydrolisis of the nitrile group, but you can look that up in your book (well known reaction). :)
Hope it helps.
Post by: stephanopolous on March 31, 2014, 09:03:02 PM
Post by: stephanopolous on March 31, 2014, 09:16:27 PM
Post by: stephanopolous on March 31, 2014, 09:35:54 PM
Hi,
In the attachment I drew the full mechanism of the first addition + elemination.
I didn't draw the hydrolisis of the nitrile group, but you can look that up in your book (well known reaction). :)
Hope it helps.
Thank you very much!!!
Post by: stephanopolous on March 31, 2014, 09:36:26 PM
So in summary of that lot:
The first product is still a nitrile.
The acid hydrolyses it to the carboxylic acid and causes the elimination of water to give the product you drew.
Thanks so much for all your help
Post by: orgopete on April 01, 2014, 05:29:40 PM
I don't think that C9H7N is correct.
Seems right to me. It is the reactants minus H2O.