Chemical Forums
Chemistry Forums for Students => Organic Chemistry Forum => Topic started by: nace17 on May 02, 2014, 02:49:01 AM
-
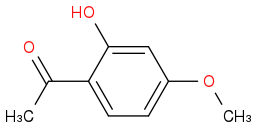
My teacher gave a molecule: benzene with a methoxy group, an alcohol group, and a ketone group. He asked why does the hydrogen in the alcohol group have a peak 12.5 ppm on the NMR. My hints were 6, water, and DNA. I have done a ton of research and haven't been able to find much of anything relating to my molecule. The only things I have found are imino protons and Watson and Crick base paring. Those are my two best guesses as of this moment. Open to any and all suggestions thank you!!
-
Here is a picture of the molecule if that helps at all.
-
1 draw your aromatic system properly! You will lose marks for that.
2 draw your bond angles properly
3 rotate the ketone group so it is closer to the OH. How do hydroxyl groups interact with carbonyls?
-
Sorry about the aromatic I had to redraw it quickly for the picture. Does the carbonyl and the hydroxyl make an acetyl?
-
No, what is one of the most fundamental properties of hydroxyl groups?
Think about your teachers comment on water
-
The polarity of the hydroxyl group. And water is also polar.
-
Any thing else? Why does water have such a high boiling point relative to methane even though their masses differ by only 2 AMU?
-
Besides bond strength and hydrogen bonding I have no idea
-
Besides bond strength and hydrogen bonding I have no idea
What would and O-H hydrogen bond to in your molecule if you rotated some bonds?
The SMILES output above gives a better conformation.
-
… only things I have found are imino protons and Watson and Crick base paring.
I am not an expert on NMR theory. I only have empirical knowledge. I presume the base paired hydrogens are shifted to low fields. Carboxylic acids are also shifted to low fields. Carboxylic acids have characteristic IR spectra presumably due to hydrogen bonding. If so, what might that tell you about your structure?
-
I was making compounds like this which had a similar phenolic proton δ=18-19 ppm is this value upfield or downfield from TMS?
What does that tell you about the proton is it sheilded or desheilded?
-
Interesting. I don't suppose you did any other studies on it, like its fractionation factor or its H-2 NMR shift? I used to do such things in a previous life.
-
Interesting. I don't suppose you did any other studies on it, like its fractionation factor or its H-2 NMR shift? I used to do such things in a previous life.
No, I have to be honest I don't know what those things are. I haven't lost the oportunity I have plenty of sample and a friendly technician who would run them. What are they?
-
When one sees downfield hydrogens one may have what are called short, strong hydrogen bonds or low-barrier hydrogen bonds. One might see that the value of the fractionation factor that is much less than one and/or a significantly nonzero value of δH - δD when comparing the two chemical shifts. The fractionation factor is a measure of whether a given exchangeable group would prefer to have hydrogen or deuterium at that site. Maurice Kreevoy did some of the early work on the organic chemistry of these H-bonds. It has been a controversial topic in enzymology. See for example, "Strong, Low-Barrier Hydrogen Bonds May Be Available to Enzymes" dx.doi.org/10.1021/bi4014566 | Biochemistry 2014, 53, 344−349
-
That's fascinating, these were only small molecules RMM = 400 ish and very unreactive at that hydroxyl group.
The hydrogen bond was so strong that you got clear HMBC data through it.
We had one which would tautomerise between two forms in chloroform give a 7:3 ratio of δ19.23 and 18.39 respectively.
How does the experiment work, you just do a simple D2O shake? or is it a bit more involved?
-
IIRC I used various ratios of H2O to D2O and took the integrals of the peak of interest. Fortunately for me, the exchange rate between bulk solvent and the peak of interest was not too fast. But that method is not applicable in all cases. I should mention that these hydrogen bonds were first found in small molecules, then enzymologists started noticing unusual fractionation factors in enzymes and reconsidering downfield signals in the serine protease enzymes. I seem to recall that Kresge and Schowen are two people who have done extensive work in the area o fractionation factors. A review article by Hibbert and Emsley might also be helpful. I'll rummage through my old papers and see what I can find. The gist of the experiment is that the heavier isotope generally enriches in the stiffer bond, and the low-barrier hydrogen bonds sit in an atypical well. I am not much of a physical organic chemist, so that is the best I can do offhand.
