Post by: curiouscat on April 19, 2016, 02:24:13 AM
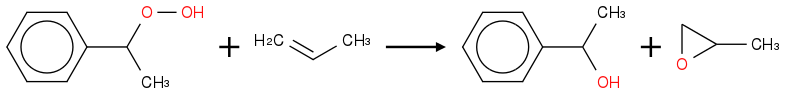
Would it be possible to use the same hydroperoxide for the following epoxidation?
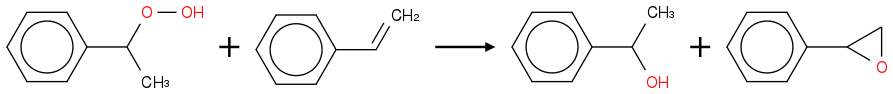
Thoughts? Is this worth a shot or likely to be a dud for fundamental reasons?
Post by: discodermolide on April 19, 2016, 02:36:49 AM
Have you checked SciFinder for this reaction to make styrene epoxide, it may well be patented.
Post by: curiouscat on April 19, 2016, 03:42:39 AM
I didn't find any blocking patents. So perhaps it just does not work well? Poor yields maybe?
Post by: orgopete on April 19, 2016, 08:08:12 AM
Post by: curiouscat on April 19, 2016, 08:28:21 AM
Is that an easy separation after the reaction is complete?
May not be "easy" but seems not onerous. BP difference is 10 C. With a vacuum this may change, though.
Experimental MP difference is 55 C! So, I suppose one could try differential crystallization but that's more messy.
Post by: curiouscat on April 19, 2016, 09:12:47 AM
I am not tied to using this one. Would any other be cheaper / work better?
Post by: discodermolide on April 19, 2016, 11:05:30 AM
Think of the equivalent naphthyl:
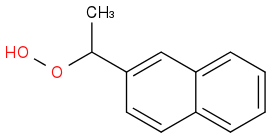
Might even be easier to separate?
Post by: Guitarmaniac86 on April 19, 2016, 12:38:06 PM
I've used it to make epoxides and to oxidise sulfur atoms in thioethers
Post by: curiouscat on April 19, 2016, 01:44:16 PM
Cumene hydroperoxide sprang into my mind. I guess hydroperoxides of similar structure would also do the same chemistry.
I considered cumene hydroperoxide too, because of its wide commercial application in Acetone / Phenol production.
But isn't cumene hydroperoxide prone to splitting up via the Hock Rearrangement? If it splits up it won't be available to epoxidate the double bond?
Post by: curiouscat on April 19, 2016, 01:54:28 PM
I've used mCPBA (meta chloro peroxybenzoic acid) for these kind of transformations because its cheap. Didn't really care if it was the most efficient or best reagent for the job, just cheap. I don't know if this is the answer you were looking for though.
Thanks! I will look into it.
Problem is one of cost: Ethylbenzene & Cumene are both available at less than $1 per kg. Air oxidation gets you the hydroperoxide.
I doubt that mCPBA can be obtained at anywhere close to this price point? Perhaps I am wrong.
Post by: curiouscat on April 19, 2016, 01:57:11 PM
Cumene hydroperoxide sprang into my mind. I guess hydroperoxides of similar structure would also do the same chemistry.
I found out that Cumene hydroperoxide is actually sold as an initiator for polymerization of Styrene. As Trigonox.
http://www.pcpds.akzonobel.com/PolymerChemicalsPDS/showPDF.aspx?pds_id=178
Ergo, we have polymerization competing with epoxidation and probably overwhelming epoxidation?
Post by: discodermolide on April 19, 2016, 02:20:13 PM
Post by: curiouscat on April 19, 2016, 02:26:48 PM
That reaction is certainly dependent upon the temperature, so you may get it to epoxidise before it polymerises?
True. It could work.
On the other hand, the Trigonox Application Note says this:
"Without accelerators effective polymerization can be obtained in the 50-200 C Range. For example, Styrene and methyl methacrylate can be polymerized in bulk at 60-100 C"
So, looks like one would have to work below at least 60 C for epoxidation selectivity to prevail. Of course, at low T rates are a different issue.