So I am trying to figure out local law regulations for the phenethylamine catch-all clause (Switzerland) and I believe they are actually the same as in the United Kingdom. I'm sure this is not against the forum rules to discuss chemical structures of drugs, I am in no point asking how to make these and my main point is to make sure that they are by definition illegal.
I don't have extended knowledge in organic chemistry so I'm not entirely sure on whether the following two cases are included by definition of the clause. I'm spent quite some time finding out the exact definitions of alkoxy, alkylenedioxy, ether, halide substituents etc and I couldn't get a definite answer yet (at least from wikipedia..). Input would be greatly appreciated if anyone would have the time xperience and enjoyment

The shortened text of the clause would be:
Any compound structurally derived from Phenethylamine, α-methylphenethylamine by substitution in the ring to any extent with alkyl, alkoxy, aklylenedioxy or halide substituents, whether or not further substituted in the ring by one or more other univalent substituents.
I include structures for phenethylamine and a-methylphenethylamine
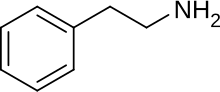
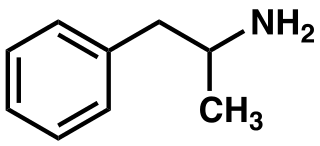
Additionally, laws in Switzerland further add this definition:
Ethers, Esters, Salts and Stereoisomers of 2C-B, DOB and para-bromoamphetamine
2C-B:

DOB:

p-bromoamphetamine:

The substances in question would be 2C-B-FLY and Bromo-DragonFLY but for simplicity I will just show the structures of them.
2C-B-FLY:

Bromo-DragonFLY:
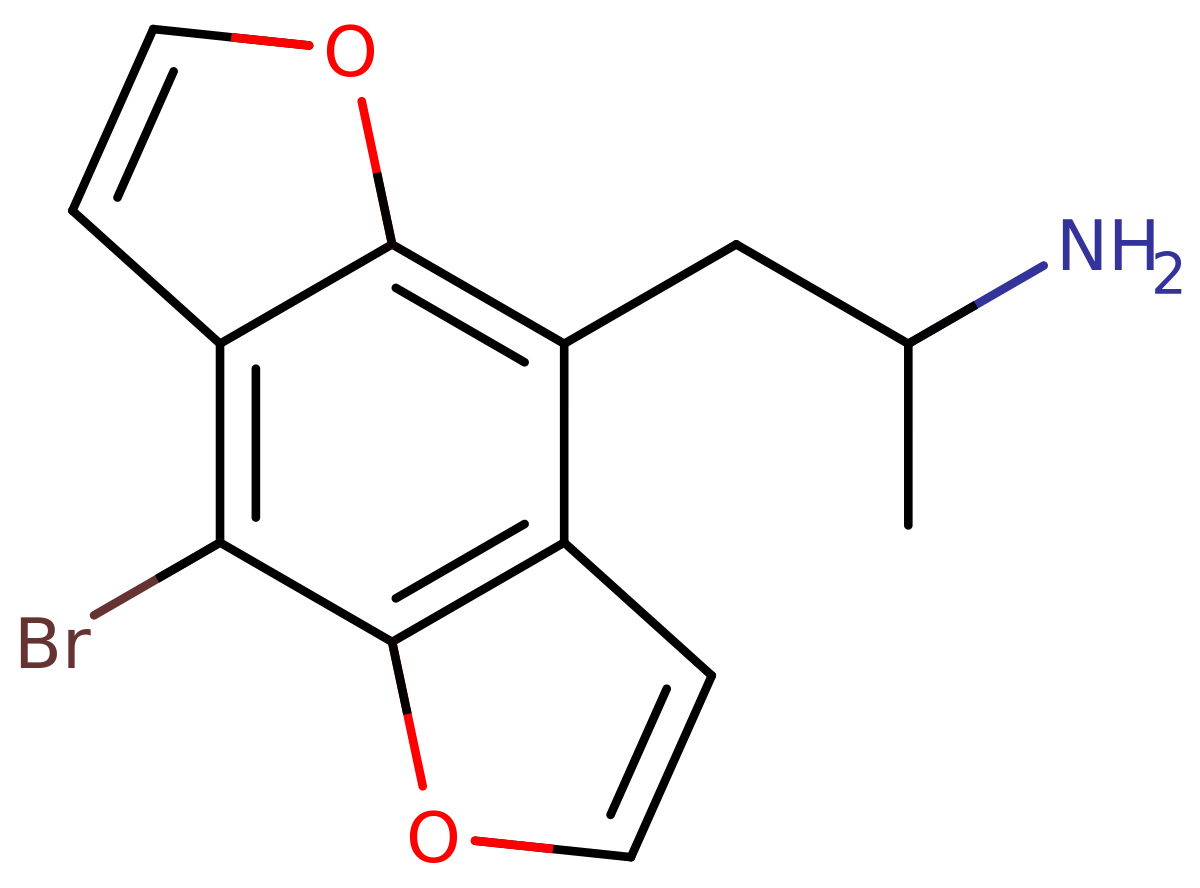
note the enclosure of the methoxy groups into a ring system bound back to the phenyl ring.
1. So 2C-B-FLY does not contain alkyl, alkylenedioxy substituents on phenethylamine. The same would go for Bromo-DragonFLY in respect to a-methylphenethylamine. I hope we can agree to this.
2. Both substances contain an halide substituent (Bromine), but the scope defines "whether or not further substituted in the ring by one or more other
univalent substituents", and I believe a dihydrofuran ring (2C-B-FLY) and furan ring (Bromo-DragonFLY) substituent is bivalent (two bonding points, which would mean it is bivalently further substituted (twice) and that is by definition not within the scope (which would only include "whether" NO or UNIVALENT substitutions)). I really hope you get me with this one.
3. One could consider the dihydrofuran rings on 2C-B-FLY as alkoxy substituents, but the alkyl part of the alkoxy group is fused with the main phenyl ring and would thereby miss an extra hydrogen where it is fused back into the ring, and is therefore not a true alkyl substituent (by definition an alkane missing just ONE hydrogen, in the case of alkoxy on the oxygen atom). Bromo-DragonFLY contains a double-bonded carbon and is therefore certainly not an alkoxy or alkane but an alkene. Maybe you could consider both as cycloalkoxy or cycloalkeneoxy?
4. Are the dihydrofuran and furan rings on 2C-B-FLY and Bromo-DragonFLY ether derivatives of 2C-B and DOB, respectively? The only two problems I'd have are first, there already being ether substitutions (this problem can be circumvented if you take p-bromoamphetamine which doesn't contain the methoxy groups. This would work for Bromo-DragonFLY), and second, the ether substitution fusing back into the ring, which would still mean that one side of the ether is not a true alkyl substituent. Rather they would be cycloethers?
Maybe I'm just overthinking this or I'm just missing something lol. Would thank anyone that takes their time