Greetings!
I am now mastering the synthesis of a polymer that I need for my work. The polymer is polymerized by step polymerization, which requires reliable chemistry.
Now for me there was a problem in stability of results. The resulting products differ in viscosity, morphology (sometimes flakes, sometimes sponge). I know that it would not be bad to characterize the molecular weight and record the ftir spectra.
Here is the reaction scheme:

In the first reaction, an isocyanate and a diol are mixed to form a polyurethane bond. After that, diamine is used for polymerization. Typically the components are added in a ratio of PCL:BDI:Putrescine 1:2:1. If we consider only the polymerization phase, that the ratio of components is 1:1. Perhaps this ratio is the problem ...
And here is the gap. The number average molecular weight can be calculated using the Carothers equation
M
w = M
0 / (1 - p)
where p is the proportion of reacted active groups.
It can be seen that if p tends to 1, then the molecular weight tends to infinity. This is impossible, but the more accurately the ratios of components are selected, the more Mn will increase, at some point being limited by the viscosity of the solution. Therefore, under conditions of exponential growth in molecular weight, any slight variations in the ratios of the components will lead to a significant change in the molecular weight of the resulting polymer.
Below is a plot of Mn versus p (Carothers equation) over the range of interest to me (Mn ~ 50,000 - 200,000).
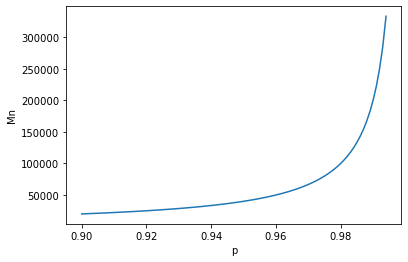
Do I understand correctly that in order to obtain a molecular weight in a given range, you need to reduce the amount of putrescine. That is, use a mixing ratio of 1:2:0.98 to get Mn ~ 100,000 (for a PCL molecular weight of 2,000). And the ratio is 1:2:0.99 for Mn ~ 200,000?