In short, no this is not true. T. McKenna has a lot of bold claims for a well studied field, I'd recommend Richard E. Schultes and Jonathan Ott for starters instead.
In length; indole is a naturally occurring molecule which is also quite commonly the parent structure of many other compounds, both natural and synthetic. One of the more notable derivatives is
tryptamine which includes things like
serotonin,
melatonin,
psilocin,
psilocybin, and by a stretch
harmala alkaloids,
ibogaine,
ergolines, many others.
Is psilocin the
only naturally 4-substituted indole? No, psilocybin,
aeruginascin,
baeocystin, and
norbaeocystin are naturally occurring and 4-substituted indoles. They are all from fungi but this doesn't mean they are necessarily anymore diverse than other sources. Certain sea creatures contain 6-halogenated tryptamines which are, to my knowledge, have never been found in plants or fungi. There are many examples of exclusive substitutions it comes down to which enzymes the are available and involved. It's not a surprise fungi create unique pattern that is limited to a handful of genera.
Indole Tryptamine 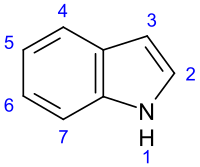

do chemists find this of interest?
It depends on the chemist but in general not likely. Jochen Gartz for example
showed addition unsubstituted synthetic substrates could be enzymatically converted into their 4-hydroxy and 4-phosphoryloxy counter parts respectively. There are many other scientists, including chemists, researching in this field ("psychedelics") but not because the 4-substitution is so unique or bizarre, which it's not compared to the diversity of nature.