I used to check the small amount of Δ+ charge on the carbonyl carbon for deciding the reactivity. If we have
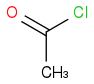
and
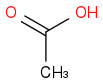
. I know that O is more electronegative than Cl and hence will increase the amount Δ+ on carbonyl carbon more than in the case of Cl also small OH molecule will provide less hinderance than large Cl. So according to me the answer should be

but the correct answer is

.Can you please explain me how? Thanks