Is it [OH]^2 = 1.2e-11/.001
Yes.
The other approach is when we try to calculate concentrations in saturated solution.
Say, we have PbCl
2 dissolving:
PbCl
2 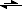
Pb
2+ + 2Cl
-and we know
Ksp=[Pb
2+][Cl
-]
How do we find how much PbCl
2 dissolved? Let's say solubility is x mol/L. That means saturated solution contains x mol/L of Pb
2+, but - because of the stoichiometry - twice as much Cl
-. So
[Pb
2+] = x
[Cl
-] = 2x
Now plug these into the Ksp and you will see where these strange coefficients come from.