ions (or ionic substances, to be more precise) can interact with water not only by being dissolved therein, but sometimes in the manner of acids / bases , additionally.
this also is true for all the ions related to phosphoric acid, where one of them ( phosphate PO
43- or "P
3-") would act as a base ( PO
43- + H
2O

HPO
42- + OH
- , pKb 1.68) exclusively, whereas the other two could act both ways:
"P2-" or hydrogenphosphate HPO42- ( pKa 12.32 ; pKb 6.79 )PO
43- + H
3O
+ 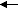
HPO
42- + H
2O

H
2PO
4- + OH
-"P-" or dihydrogenphosphate H2PO4- ( pKa 7.21 ; pKb 11.84 )HPO
42- + H
3O
+ 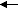
H
2PO
4- + H
2O

H
3PO
4 + OH
-As you see, in P
2- both these properties kinda "fight against each other". However, the base - properties being stronger by the factor of approx. 10 to the power of five, this effect will prevail, and therefore solutions of hydrogenphopsphate will end up being alkaline ( pH approx. 9.77 , to be more specific)
regards
Ingo