Hello Dan, I tried to use the SMILES but it didn't work for some reason. But I have looked at what you typed and it should be fine now.
Which of the following compounds react with acetic acid? What are the products?
So my idea is that the O and the OH in the acetic acid will react since it is slightly negative as it draws the electrons to itself.
Phenol:
Phenol + acetic acid --> ester + water
Aniline:
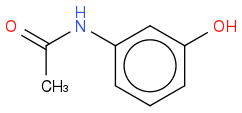
+ water
Nitrobenzene
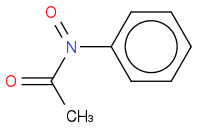
+ water
Bromobenzene
I doubt it reacts but the best I could come up with was this:
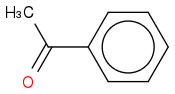
+ BrOH
Toluene
Not sure about this
My reactions are all based on my knowledge of organic chem, I am probably wrong. I have tried looking for sites that give you a general idea to predict organic reactions but can't find any.
Also, is the O or the OH in acetic acid more reactive? Or does it depend?
Thanks